| Research Article | ||
J. Microbiol. Infect. Dis., (2025), Vol. 16(1): 18–22 Research Article Identification of gastrointestinal parasites and their effects on blood parameters in free-range chickensNagia A. S. Abdalsalam1*, Hana A. Ali2 and Ahmad Boulabyidh21Department of Pathology and Clinical Pathology, Faculty of Veterinary Medicine, Omar Al-Mukhtar University, Al-Bayda, Libya 2Department of Parasitology, Faculty of Veterinary Medicine, Omar Al-Mukhtar University, Al-Bayda, Libya *Corresponding Author: Nagia A. S. Abdalsalam. Department of Pathology and Clinical Pathology, Faculty of Veterinary Medicine, Omar Al-Mukhtar University, Al-Bayda, Libya. Email: nagia.abdalsalam [at] omu.edu.ly Submitted: 25/10/2025 Revised: 03/12/2025 Accepted: 08/12/2025 Published: 01/01/2026 © 2026 Open Veterinary Journal
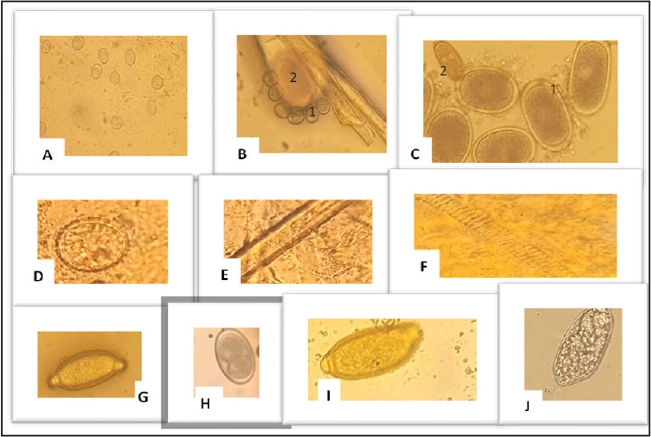
ABSTRACTBackground: Gastrointestinal (GIT) parasites can reduce poultry productivity by negatively affecting the health of birds and altering specific blood values. Few published studies have addressed this issue in Libya, particularly in Al-Bayda and the surrounding areas. Aim: This study aimed to evaluate the presence and impact of GIT parasites on selected hematological parameters in selected free-range chickens (crossbreed chicken, Libyan chicken, and Fyomi) in Al-Bayda and the surrounding areas of Al-Bayda in Libya. Methods: A total of 50 fecal samples from adult chickens collected from local poultry breeders were examined for GIT parasites using the flotation technique. Blood parameters, including RBC, hemoglobin, packed cell volume, mean corpuscular volume, mean corpuscular hemoglobin, mean corpuscular hemoglobin concentration, and WBC, were also collected and analyzed using an automated hematology analyzer (Mindray). Statistical significance between complete blood count parasite positivity and reference parameters was determined using the mean standard deviation (mean ± SD). Results: We detected GIT parasites in 100% of the samples. Among these, 48% had a single infection, 36% had two different parasites, and 16% had three parasites. The common parasites identified included Ascaridia spp. (24%), Heterakis spp. (14%), Capillaria spp. (18%), Eimeria spp. (64%), Trichostrongylus (12%), and Choanotaenia (20%). Several factors were significantly associated with the presence of GIT parasites, including farm location, years of operation, number of employees, proximity to water sources, fecal cleaning practices, and other animal presence. Conclusion: GIT parasites were found in the poultry of breeders in selected areas of Al-Bayda, Libya. These findings highlight the need for regular monitoring of GIT parasites and emphasize the need for consistent deworming or rotation of dewormers in the region. Keywords: Gastrointestinal parasites, Free-range chickens, Blood parameters, Libya. IntroductionPoultry meat is one of the most widely consumed animal-derived foods globally. Chickens provide essential resources for crop fertilization, including eggs, meat, and manure (Wamboi et al., 2020). According to the Food and Agriculture Organization (2009), poultry meat accounts for approximately 30% of the global animal protein consumed by humans. Breeding farm animals is crucial for food production and income generation in rural Libyan communities. The poultry sector is crucial for national economic development, as poultry complexes fulfill over 40% of Libya's chicken meat requirements (Guèye, 2004). However, information regarding the health of these chickens is scarce. From a health perspective, free-range chickens may play a role in transmitting diseases to humans. However, the surveillance and diagnosis of infections, such as parasitic diseases, that impact the productivity of these chickens are often limited to postmortem examinations (Cantin-Rosas et al., 2025). Additionally, information about the overall health of free-range chickens is lacking. The gastrointestinal (GIT) tract is often exposed to infections from various pathogenic organisms, especially parasitic helminths, which can absorb nutrients and increase the risk of other diseases. These factors pose a significant threat to the poultry industry, leading to economic losses, reduced production performance, and a lower feed conversion ratio. These issues can cause numerous health problems in birds, including decreased egg production, catarrh, loss of appetite, diarrhea, intestinal blockage, emaciation, anemia, weakness, paralysis, poor feathering, and even death (Makouloutou-Nzassi et al., 2024). GIT helminths, such as Heterakis gallinarum, can transmit pathogens, such as Histomonas meleagridis, in turkeys and chicks (Opara et al., 2014), resulting in high morbidity and up to 20% mortality in chicken flocks (Makouloutou-Nzassi et al., 2024). Although Angiostrongylus cantonensis can cause mild neurological problems in domestic chickens, these birds are not particularly susceptible to infection by this parasite (Fecková et al., 2021). In addition, numerous nematodes and cestode parasites infect free-range chickens, with over 30 helminth species identified in domestic chickens. These include nematodes (roundworms) such as Ascaridia galli, Heterakis spumosa, Capillaria species, Trichostrongylus, and parasites such as Choanoteania (Cantin-Rosas et al., 2025). They result in significant illness and death, leading to considerable economic losses for the poultry industry (Singh et al., 2023). These parasites damage the intestinal tract, causing malabsorption, malnutrition, anemia, emaciation, bleeding, weight loss, and reduced egg production (Shohana et al., 2023). Infection can occur due to various species of Eimeria spp., each exhibiting different levels of pathogenicity and associated with production losses (Cantin-Rosas et al., 2025). Blood parameters are valuable indicators of the health status, nutritional needs, and physiological conditions of animals (Zálešáková et al., 2025), but they are frequently underutilized (Wamboi et al., 2020). Monitoring changes in blood parameters can aid in the early diagnosis of poultry parasites and reduce production losses (Zálešáková et al., 2025). In Libya, no studies have evaluated the clinicopathological parameters used for diagnosing avian diseases. Therefore, this study aimed to identify and diagnose helminths, including cestodes, nematodes, and trematodes, and to assess their impact on specific blood parameters in domestic chickens. Materials and MethodsSample sizeTo calculate the sample size for comparing a blood parameter (continuous variable) between two independent groups (e.g., infected vs. uninfected chickens), the appropriate formula is the sample size for comparing two means: n: sample size per group Z1 -α/2: Z-value for the desired confidence level (e.g., 1.96% for 95% CI) Z1−β: Z-value for the desired power (e.g., 0.84% for 80% power) Expected standard deviation (σ): 1.5 g/dl Expected difference (d): 1.0 g/dl Hence, about 18 samples per group. Approximately 50 samples were collected to avoid any loss due to dead chickens or invalidation of the chicken samples. AnimalsThis study was conducted from June to August 2025, involving 50 adult chickens of both sexes aged between 1 and 3 years. Chickens were sourced from breeders and housed overnight in cages at the Omar Al-Mukhtar University Veterinary Clinic. Collection and analysis of fecal samplesThe chickens were brought from the breeders and placed in cages at the Omar Al-Mukhtar University Veterinary Clinic overnight. In the morning, one fecal sample was collected rectally from each chicken in sterile bags. A flotation test was performed on the feces (Conboy, 2025) to detect the presence of helminth eggs according to the method described by Kelly (1974). The investigation was limited to the use of fecal samples; necropsy was not performed to recover worms. Blood samplesBlood samples were collected from the wing vein into a clean test tube containing ethylenediaminetetraacetic acid for plasma, labeled, and sent to the laboratory. Blood parameters, RBC, hemoglobin (Hb), packed cell volume (PCV), mean corpuscular volume (MCV), mean corpuscular hemoglobin (MCH), mean corpuscular hemoglobin concentration (MCHC), and WBC were examined using the automated hematology analyzer (Mindray). Statistical analysisData were entered and analyzed using SPSS, version 26, and expressed as mean ± standard deviation (mean ± SD). Ethical approvalThis study was approved by the Al-Mukhtar Committee for Bio-safety and Bioethics at Omar Al-Mukhtar University. The approval number is NBC: 007. A. 25. 54. ResultsIn this study involving 50 chickens, 100% was found to carry at least one genus of parasites. Specifically, 48% had one genus, 36% had two genera, and 16% had three genera. The seven parasitic genera and species detected included Eimeria spp. (64%), A. galli (24%), H. gallinarum (14%), Capillaria spp. (18%), Trichostrongylus (12%), and Choanotaenia (20%) (Fig. 1).
Fig. 1. Intestinal parasites found in free range chicken (Fuome) brough from the breeders. Following flotation analysis, the samples were seen at 400 m magnification using a light microscope. The pictures depict eggs (C,D) or oocysts (A from the parasites listed below: (A) Eimeria spp, (B))1- Eimeria spp and 2-A. galli, (C) 1-A. galli and 2-Capillaria spp (D) oocyst of choanoteania, (E +F Tpwarm of choanoteania, (G) Trichuris spp. (H) H. gallinarum, (I) Capillaria spp and (J) Trichostrongylus spp. Clinical examinations of infected chickens revealed that most were suffering from anemia, diarrhea, emaciation, pale mucous membranes, and stunted growth. Hematological analysis of blood samples collected from these infected chickens showed a significant reduction in Hb, total erythrocyte counts (RBC), PCV values, MCV, MCH, and MCHC. In contrast, a significant increase leukocyte count (WBC) was observed compared with control chickens (Table 1). All the examined chickens tested positive for intestinal parasitic eggs using qualitative methods. Table 1. Comparison of the results of some hematological parameters with reference values (mean + SD) from previous studies and the source of the reference values. Notable deviations were observed when comparing these parameters with established reference intervals from prior studies. GIT parasites caused the following reductions (mean ± SD): RBC (2.62 ± 0.39), PCV (23.4 ± 4.2), Hb (6.78 ± 2.54), MCV (104.16 ± 8.63), MCH (22.13 ± 7.54), and macrophage colony count (25.12 ± 2.04). Conversely, the WBC count increased (33.98 ± 1.02). Differences were statistically significant (p ˂ 0.05). DiscussionThis study revealed a 100% prevalence of GIT parasites in the examined chickens, with mixed infections identified. The most prevalent parasite was Eimeria spp. (64%), followed by cestodes, including Raillietina spp. and Choanotaenia spp., and nematodes, such as A. galli and Capillaria spp. These findings align with reports indicating that free-range poultry are at a higher risk of parasitic infestation due to continuous exposure to contaminated soil, water, and fecal matter (Alsaad, 2016; Conboy et al., 2025). The overall prevalence was nearly 100%, consistent with findings from Nigeria, where almost all local chickens (100%) were reported to be infected with GIT parasites (Alsaad, 2016). This result also corresponds with findings reported from Ethiopia, which documented a prevalence of 90.60% (Udensi et al., 2019). In contrast, the results of this study were higher than those reported by Berhe et al. (2019) and Kumari et al. (2024). The high prevalence observed in this study may be attributed to favorable climatic conditions that promote helminth development. In addition, free-range chickens forage on the ground and may consume intermediate hosts, such as insects and other invertebrates, including ants and earthworms, which can act as vectors (Rahman et al., 2024). In previous studies, similar GIT parasites, including Eimeria spp., Ascaridia spp., Heterakis spp., Capillaria spp., and Raillietina sp., have been identified in Nigeria, Ethiopia, Bangladesh, and Thi-Qar province (Berhe et al., 2019; Udensi et al., 2019; Kumari et al., 2024; Rahman et al., 2024). The hematological results showed significant reductions in the red blood cell count (RBC), Hb concentration, PCV, MCV, MCH, and MCHC. In contrast, the total WBC count was significantly elevated. This pattern is characteristic of parasitic anemia and an immune response to infection. Similar findings were observed in experimentally infected broilers with Eimeria spp. (Tamirat and Hassan, 2024) and in rural chickens exposed to intestinal parasites (Rom et al., 2021). The reduction in erythrocytic indices may result from several mechanisms. Direct blood loss and tissue damage from helminths, such as A. galli and Capillaria spp., can compromise intestinal integrity as these parasites consume host tissues (Parveen et al., 2017). Additionally, nutrient malabsorption, particularly due to mucosal damage caused by Eimeria spp., leads to impaired erythropoiesis and reduced Hb synthesis (Ebrahim, 2018). Chronic stress and inflammation can further suppress bone marrow activity, thereby exacerbating anemia (Opara et al., 2014). The significant increase in WBC counts observed in infested chickens indicates the activation of both innate and adaptive immunity. This finding aligns with previous research conducted on sheep and poultry, which reported elevated WBC counts as a defense mechanism against parasitic infections (Al-amery, 2018; Rom et al., 2021). The rise in leukocyte counts reflects the recruitment of immune cells to the inflamed intestinal mucosa, a key feature of parasitic pathogenesis. The clinical manifestations observed in this study—such as anemia, diarrhea, pale mucous membranes, emaciation, and reduced growth performance—are consistent with descriptions found in parasitology literature and diagnostic studies (Uhuo et al., 2013; Alsaad, 2016; Conboy et al., 2025). These symptoms, along with the observed hematological changes, provide compelling evidence that GIT parasites harm poultry's overall health and productivity. These findings indicate that hematological parameters can serve as effective diagnostic markers of parasitic infections in chickens. This aligns with previous reports that emphasized the use of complete blood count as a reliable indicator for monitoring livestock health and adaptive responses (Rick et al., 2004; Ibrahim, 2021). ConclusionThis study emphasizes the importance of regular parasitological screenings, strategic deworming programs, improved farm hygiene, and enhanced management practices. Implementing these measures is crucial for reducing the negative impact of GIT parasites on poultry productivity in Libya and other similar environments. To the best of the authors' knowledge, this is the first documented report of hematological alterations associated with GIT parasites in free-range chickens from eastern Libya. AcknowledgmentsThe authors gratefully acknowledge the support and cooperation of the Universitas Omer Al-Mukhtar. Authors’ contributionsAll authors contributed to making the completion of this manuscript possible. Collecting samples and conducting the research trials. Classification of the parasite. Data curation. Writing original draft preparation: Finishing the manuscript. ReferencesAl-amery, A. 2018. Effect of Ascaridia galli infection on some blood parameters in layer chickens. Mirror of Research in Veterinary Sciences and Animals (MRVSA), Basrah, Iraq. Alsaad, K.M. 2016. Veterinary clinical pathology: procedures for domesticated animals. University of Basrah Press, Basrah, Iraq, 6–164. Berhe, M., Mekibib, B., Bsrat, A. and Atsbaha, G. 2019. Gastrointestinal parasites of chickens under different management systems in Mekelle, Northern Ethiopia. J. Vet. Med. 2019, 1307582; doi:10.1155/2019/1307582 Cantin -Rosas, B., Tomazic, M.L., Rodríguez, A.E., Enciso, N., Brante-Bernier, J., Honores, P, et al. 2025. Risk factors and spatial distribution of gastrointestinal parasites in chickens. Vet. Sci. 12(5), 1–15. Conboy, G. 2025. Parasitology in veterinary practice [Internet]. In Merck veterinary manual internet. Ed., Abuelo, A. Rahway, NJ: Merck & Co., Inc. Available via https://www.msdvetmanual.com/clinical-pathology-and-procedures/parasitology/parasitology-in-veterinary-practice Ebrahim, Z. 2018. Effect of gastrointestinal parasitic infestation on hematological and biochemical parameters in chickens. Alexandria J. Vet. Sci. 59(1), 44; doi:10.5455/ajvs.1922 Fecková, B., Hurníková, Z. and Miterpáková, M. 2021. Angiostrongylus cantonensis in domestic chickens. Parasitol. Res. 120(8), 2891–2895. Food and Agriculture Organization. 2009. The state of food and agriculture 2009. Rome, Italy: FAO. Guèye, E.F. 2004. The role of family poultry in poverty alleviation, food security and the promotion of gender equality in rural Africa. Outlook Agric. 33(4), 259–266. Iacob, S., Iacob, R., Manea, I., Uta, M., Chiosa, A., Dumbrava, M. and Popescu, I. (2021). Establishment of local hematological reference intervals. Journal of Blood & Lymph, 11(5). Ibrahim, A.A. 2021. Hematological and biochemical parameters of indigenous chickens in Nigeria. Anim. Prod. Res. Adv. 5, 51–68. Kelly, J.D. 1974. A modification of the zinc sulphate flotation technique. Austral. Vet. J. 50(10), 472. Kumari, P., Rahman, S. and Ahmed, N. 2024. Prevalence and identification of gastrointestinal parasites in domestic chickens. Vet. World 17(3), 458–463. Makouloutou-Nzassi, P., Bouyer, J., Ravel, S. and Mediannikov, O. 2024. Gastrointestinal parasites of chickens in central Africa. Parasit. Vectors. 17, 123. Opara, M.N., Osowa, D.K. and Maxwell, J.A. 2014. Blood and gastrointestinal parasites of chickens from selected farms in Owerri, Southeast Nigeria. Open J. Vet. Med. 4(12), 308–313. Parveen, A., Khan, M.H., Khawaja, T., Iftikhar, N. and Khan, S. 2017. Growth performance and hemato- biochemical parameters of different chicken breeds. J. World. Poult. Res. 7(3), 114–122. Rahman, M.A., Hossain, M.S. and Islam, K. 2024. Gastrointestinal parasites of chickens in selected areas of Bangladesh. Bangladesh J. Vet. Med. 22(1), 67–73. Rick, L., Alleman, A.R., Cunningham, D.J., Deheer, H.L. and Dorsey, K. 2004. Veterinary clinical pathology secrets. Philadelphia, PA: Hanley & Belfus; doi:10.1016/b978-1-56053-633-8.50001-2 Rom, R., Ciornohac, M. and Pavel, G. 2021. Haematobiochemical parameters in broiler chickens experimentally infected with Eimeria spp in variable chemoprophylaxis and therapy conditions. Rom. J. Vet. Med. 31(1), 69–76. Shohana, S. S., Islam, M. S., Sikder, S. and Rahman, M. M. 2023. Ascaridia galli infection in chicken: pathobiology and immunological orchestra. Immun. Inflamm. Dis. 11(9), 1–11; doi:10.1002/iid3.1001 Singh, R., Gupta, I. and Patil, R.D. 2023. Ascariasis in poultry: a comprehensive review. Pharma. Innov. 12(11S), 699–704. Tamirat, H. and Hassan, M. 2024. Epidemiological study of gastrointestinal parasites in backyard chickens. Vet. Parasitol. 325, 110089. Udensi, A.U., Ugbogu, P. and Ekesiobi, U.E. 2019. Prevalence of gastrointestinal helminths in local and broiler chickens in Ibesikpo Local Government Area, Akwa Ibom State, Nigeria. Am. J. Zool. Res. 7(1), 1–7; doi:10.12691/ajzr-7-1-1 Uhuo, A.C., Okafor, F.C., Odikamnoro, O.O., Onwe, C.S., Abarike, M.C. and Elom, J.U. 2013. Common gastrointestinal parasites of local chicken (Gallus domesticus) slaughtered in some selected eatery centres in Abakaliki, Ebonyi State: implication for meat quality. Int. J. Dev. Sustain. 2(2), 1416–1422. Wamboi, P., Waruiru, R.M., Mbuthia, P.G., Nguhiu, J.M. and Bebora, L.C. 2020. Haemato-biochemical changes and prevalence of parasitic infections of indigenous chicken sold in markets of Kiambu County, Kenya. Int. J. Vet. Sci. Med. 8(1), 18–25; doi:10.1080/23144599.2019.1708577 Zálešáková, D., Novotný, J., Řiháček, M., Horáková, E., Mrkvicová, E. and Šťastník, O. 2025. Hematological and biochemical parameters, reference intervals, and dynamics in modern broiler chickens. Vet. Anim. Sci. 29, 100465; doi:10.1016/j.vas.2025.100465 | ||
| How to Cite this Article |
| Pubmed Style Abdalsalam NAS, Ali HA, Boulabyidh A. Identification of gastrointestinal parasites and their effects on blood parameters in free-range chickens. J Microbiol Infect Dis. 2026; 16(1): 18-22. doi:10.5455/JMID.2026.v16.i1.3 Web Style Abdalsalam NAS, Ali HA, Boulabyidh A. Identification of gastrointestinal parasites and their effects on blood parameters in free-range chickens. https://www.jmidonline.org/?mno=292598 [Access: January 25, 2026]. doi:10.5455/JMID.2026.v16.i1.3 AMA (American Medical Association) Style Abdalsalam NAS, Ali HA, Boulabyidh A. Identification of gastrointestinal parasites and their effects on blood parameters in free-range chickens. J Microbiol Infect Dis. 2026; 16(1): 18-22. doi:10.5455/JMID.2026.v16.i1.3 Vancouver/ICMJE Style Abdalsalam NAS, Ali HA, Boulabyidh A. Identification of gastrointestinal parasites and their effects on blood parameters in free-range chickens. J Microbiol Infect Dis. (2026), [cited January 25, 2026]; 16(1): 18-22. doi:10.5455/JMID.2026.v16.i1.3 Harvard Style Abdalsalam, N. A. S., Ali, . H. A. & Boulabyidh, . A. (2026) Identification of gastrointestinal parasites and their effects on blood parameters in free-range chickens. J Microbiol Infect Dis, 16 (1), 18-22. doi:10.5455/JMID.2026.v16.i1.3 Turabian Style Abdalsalam, Nagia A. S., Hana A. Ali, and Ahmad Boulabyidh. 2026. Identification of gastrointestinal parasites and their effects on blood parameters in free-range chickens. Journal of Microbiology and Infectious Diseases, 16 (1), 18-22. doi:10.5455/JMID.2026.v16.i1.3 Chicago Style Abdalsalam, Nagia A. S., Hana A. Ali, and Ahmad Boulabyidh. "Identification of gastrointestinal parasites and their effects on blood parameters in free-range chickens." Journal of Microbiology and Infectious Diseases 16 (2026), 18-22. doi:10.5455/JMID.2026.v16.i1.3 MLA (The Modern Language Association) Style Abdalsalam, Nagia A. S., Hana A. Ali, and Ahmad Boulabyidh. "Identification of gastrointestinal parasites and their effects on blood parameters in free-range chickens." Journal of Microbiology and Infectious Diseases 16.1 (2026), 18-22. Print. doi:10.5455/JMID.2026.v16.i1.3 APA (American Psychological Association) Style Abdalsalam, N. A. S., Ali, . H. A. & Boulabyidh, . A. (2026) Identification of gastrointestinal parasites and their effects on blood parameters in free-range chickens. Journal of Microbiology and Infectious Diseases, 16 (1), 18-22. doi:10.5455/JMID.2026.v16.i1.3 |