| Case Report | ||
J. Microbiol. Infect. Dis., (2026), Vol. 16(1): 37–43 Case Report Multiple cerebral abscesses caused by Aspergillus fumigatus following extracorporeal membrane oxygenation in a pediatric patient: A case reportChutima Phuaksaman1, Nuanluck Yupensuk1, Veeratape Ngamnusonkit1, Krittaporn Phruksarudee1, Julintorn Somran2, Jiroje Jiranukool3 and Sarunya Srijuntongsiri1*1Department of Pediatrics, Faculty of Medicine, Naresuan University, Phitsanulok, Thailand 2Department of Pathology, Faculty of Medicine, Naresuan University, Phitsanulok, Thailand 3Department of Surgery, Faculty of Medicine, Naresuan University, Phitsanulok, Thailand *Corresponding Author: Sarunya Srijuntongsiri. Department of Pediatrics, Faculty of Medicine, Naresuan University, Phitsanulok, Thailand. Email: sarunyas [at] nu.ac.th, sarnyachin [at] gmail.com Submitted: 24/10/2025 Revised: 22/01/2026 Accepted: 05/02/2026 Published: 03/03/2026 © 2026 Open Veterinary Journal
ABSTRACTBackground: Invasive cerebral aspergillosis is a rare but serious condition with a high mortality rate. Case Description: We report a case of multiple brain abscesses with ventriculitis caused by Aspergillus fumigatus in an 11-year-old Thai boy who presented with a high-grade fever and progressive headache for 9 days during hospitalization. He was diagnosed with severe acute respiratory distress syndrome and secondary hemophagocytic lymphohistiocytosis. Treatment included veno-arterial-venous extracorporeal membrane oxygenation, broad-spectrum antibiotics, and prolonged corticosteroid therapy. He subsequently developed severe headaches due to increased intracranial pressure and brain edema, which were successfully managed with a combination of antifungal therapy and extensive neurosurgery. Conclusion: Early diagnosis and combined antifungal therapy, in combination with neurosurgery, are crucial for improving survival outcomes in invasive cerebral aspergillosis. Keywords: Brain abscess, Aspergillosis, ECMO. IntroductionInvasive cerebral aspergillosis is a rare disease with a high morbidity and mortality rate exceeding 80% in immunocompromised patients (Dotis et al., 2007). Patients undergoing immunosuppressive therapy for bone marrow or solid organ transplantation, hematologic malignancies, or hemophagocytic lymphohistiocytosis, as well as those receiving prolonged corticosteroids or with chronic granulomatous disease, are at particularly high risk (Sullivan et al., 2022). Cerebral aspergillosis typically arises from hematogenous dissemination from the lungs or invasive sinus infection and usually manifests as single or multiple brain abscesses, rarely as ventriculitis or meningitis (McCarthy et al., 2014; McCarthy et al., 2017). Few cases of cerebral aspergillosis have been described in children, with rare favorable outcomes. Here, we report a case of multiple cerebral abscesses caused by Aspergillus fumigatus in a child with severe acute respiratory distress syndrome (ARDS) and secondary hemophagocytic lymphohistiocytosis (sHLH) during steroid treatment following veno-arterial-venous extracorporeal membrane oxygenation (VAV-ECMO). Case DescriptionAn 11-year-old Thai boy with a history of untreated asthma presented with high-grade fever and a productive cough for 5 days. He was admitted to a provincial hospital with a diagnosis of pneumonia and asthmatic exacerbation. Five hours post-admission, he developed respiratory failure requiring intubation. His clinical condition continued to deteriorate, characterized by profound desaturation and bilateral patchy infiltrates on chest X-ray, consistent with ARDS. Initial treatment included ceftriaxone, oseltamivir, azithromycin for pneumonia, and hydrocortisone. He subsequently developed hypotension and was treated with adrenaline, dobutamine, intravenous immunoglobulin, and pulse methylprednisolone for 2 days before being transferred to Naresuan University Hospital (NUH), Phitsanulok, Thailand, due to worsening ARDS. Upon arrival at NUH, the patient’s vital signs were: body temperature 38°C, RR 30 bpm, HR 164 bpm, BP 93/39 mmHg, and SpO2 66–73% on mechanical ventilation (FiO2 1.0). Physical examination revealed fine crepitations in both lungs and hepatosplenomegaly. Laboratory investigations showed pancytopenia, elevated ferritin levels, bone marrow aspiration demonstrating hypocellularity, and increased histiocytes with hemophagocytosis, consistent with secondary hemophagocytic lymphohistiocytosis (sHLH) (Table 1). Blood, sputum, urine, and bone marrow cultures were negative. Empirical therapy with meropenem, vancomycin, and dexamethasone 10 mg/m2/day was initiated. Table 1. Laboratory examination results.
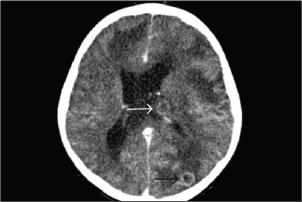
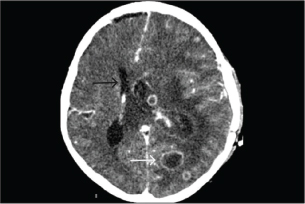
Despite optimized conventional mechanical ventilation, oxygenation remained poor, with an oxygenation index of 85. VAV-ECMO was therefore initiated for severe ARDS and myocarditis with shock. VAV-ECMO support was maintained for 8 days, and the patient was successfully extubated after 12 days of hospitalization. Repeated sputum culture, hemoculture, and 33 respiratory pathogen panels remained negative, and follow-up chest radiography demonstrated resolution of pulmonary infiltrates. One-week post-VAV-ECMO, while still on corticosteroids for 15 days, the patient developed a new high-grade fever that persisted for 9 days. On day 23 of admission, he complained of progressive headache and agitation. Neurological examination revealed bilaterally dilated (5 mm) but reactive pupils without focal deficits. Emergency contrast-enhanced computed tomography (CT) of the brain demonstrated multiple cerebral abscesses with vasogenic edema and one lesion located at the left periventricular area, adjacent to the lateral ventricle (Fig. 1). In immunocompetent individuals, the most common pathogens were Streptococcus spp., Staphylococcus aureus, Enterobacteriaceae, Pseudomonas aeruginosa, and anaerobes. In immunocompromised hosts, it is most commonly caused by toxoplasmosis, Nocardia spp., Candida spp., Aspergillus spp., and Cryptococcus spp. Empirical antimicrobial therapy was adjusted to intravenous meropenem and vancomycin to provide coverage for multidrug-resistant bacteria and methicillin-resistant Staphylococcus aureus (MRSA). A right frontal craniotomy was subsequently performed for abscess drainage and diagnostic confirmation, along with placement of an external ventricular drain (EVD) in the left lateral ventricle for cerebrospinal fluid (CSF) sampling and intracranial pressure control.
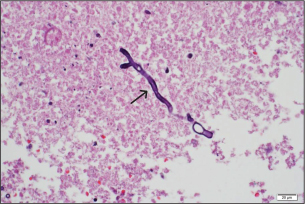
Fig. 1. A computed tomography scan of the brain with contrast on day 23 of admission showed two ring-enhancing lesions located in the left occipital lobe (black arrow) and left periventricular region, adjacent to the lateral ventricle (white arrow). KOH examination of the abscess material demonstrated septate hyphae. Histopathological analysis of the brain abscess from the right frontal lobe revealed uniform, dichotomously branching septate hyphae with acute-angle branching infiltrating the vascular wall and surrounding brain parenchyma (Fig. 2). CSF analysis revealed hypoglycorrhachia, elevated protein, and neutrophilic pleocytosis (Table 1). Cryptococcal antigen testing was negative. There was no radiologic evidence of disseminated aspergillosis involving the lungs or paranasal sinuses. Extensive microbiological investigations for aerobic and anaerobic bacteria, Mycobacterium tuberculosis, Nocardia spp., herpes simplex virus, cytomegalovirus, and Toxoplasma gondii were negative. Serum galactomannan testing showed 0.42; CSF galactomannan and β-D-glucan assays were not performed. These findings supported a diagnosis of multiple cerebral abscesses caused by aspergillosis without evidence of extracranial dissemination.
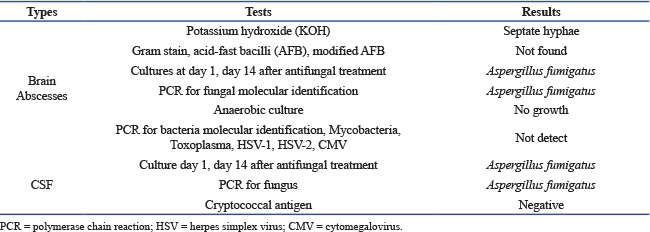
Fig. 2. Brain abscess, Hematoxylin and eosin (H&E) stain, 40×: on day 23 of admission, Fungal hyphae typically branch at acute angles in the necrotic area (black arrow). The patient was started on intravenous voriconazole with therapeutic drug monitoring (9 mg/kg/dose q 12 hours for the first two doses as a loading regimen, followed by 8 mg/kg/dose q 12 hours as maintenance therapy) in combination with amphotericin B deoxycholate (1 mg/kg/day), meropenem, and a steroid taper. Anticonvulsants (phenytoin), electrolyte correction, pain control, and supportive treatment for brain edema were provided. The patient became afebrile 3 days after initiation of antifungal therapy and abscess removal. One week after surgery, follow-up brain CT showed enlargement of multiple ring-enhancing lesions in the left frontal, parietal, and occipital lobes, left lateral ventriculitis (Fig. 3). Intraventricular amphotericin B deoxycholate 0.5 mg once daily was added to his antifungal regimen for 14 days due to a lack of radiologic response to ongoing therapy. The fungal isolate was identified as Aspergillus fumigatus on Sabouraud dextrose agar media of abscess and CSF; greenish, powdery, non-velvety colonies and lactophenol cotton blue staining showed septate hyphae, dichotomous branching, columnar conidial head, flask-shaped vesicle, limited phialide upper half of two-thirds of the vesicle, and uniseriate conidia head. Polymerase chain reaction (PCR) analysis of the brain abscess confirmed Aspergillus fumigatus, while bacterial cultures and PCR were negative (Table 2). HIV testing was negative, and lymphocyte subset analysis (CD3, CD4, and CD8) and serum immunoglobulin levels (IgA, IgE, IgG, and IgM) were within normal ranges. Neutrophil oxidative burst testing (NBT) or Dihydrorhodamine test (DHR) and lymphocyte proliferation assays were deferred, as these tests should be performed at least 1 month after blood transfusion to ensure accurate results.
Fig. 3. A computed tomography scan of the brain with contrast on day 23 of admission showed two ring-enhancing lesions located in the left occipital lobe (black arrow) and left periventricular region, adjacent to the lateral ventricle (white arrow). Table 2. Microbiological results.
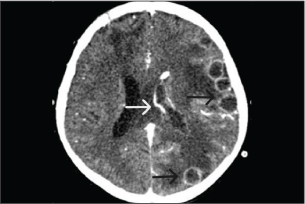
The voriconazole trough concentration on day 9 of treatment was 0.11 mg/l. Concomitant elevation of liver enzyme levels was observed, prompting discontinuation of phenytoin and substitution with levetiracetam, while the voriconazole dose was maintained. After 3 weeks of combination antifungal therapy, a brain CT scan demonstrated a stable abscess size with a slight increase in periventricular hypodensity in the left temporal lobe. A second craniotomy was performed to excise the largest abscesses in the bilateral frontal lobes to relieve mass effect, with continuation of intravenous voriconazole and amphotericin B. A repeat voriconazole trough concentration measured 1.9 mg/l, within the target therapeutic range of 1–5.5 mg/l. Liver enzyme levels had returned to normal. On day 28 of combination antifungal therapy, the patient experienced a severe left temporal headache accompanied by motor weakness (grade III on the right side and grade IV on the left side). Repeat brain CT demonstrated progression of abscesses, left lateral ventriculitis, increased vasogenic edema, and uncal herniation (Fig. 4). The patient was deteriorating, and there was significant brain swelling. A third neurosurgical intervention involving left decompressive craniectomy with duraplasty and EVD placement was performed to relieve intracranial pressure. Postoperatively, the patient showed slight improvement in motor function and maintained normal consciousness.
Fig. 4. A computed tomography scan of the brain with contrast on day 28 of antifungal treatment showed increased size of some abscesses (white arrow), ventriculitis of the left lateral ventricle, increased vasogenic edema, and midline shift to the right (black arrow). Amphotericin B was discontinued after 1 month, and intravenous voriconazole was switched to oral voriconazole (200 mg twice daily). The patient was transferred back to the provincial hospital for continuation of intravenous meropenem for 6 weeks, oral voriconazole therapy, and rehabilitation. Functional outcome was assessed using the Glasgow Outcome Scale; however, formal scoring was not documented. Follow-up assessments were conducted via telephone communication with the patient’s grandmother and local physician due to difficulty attending in-person visits. DHR testing was planned once he was clinically stable and able to return to the hospital for follow-up evaluation. The patient was discharged with residual motor weakness. Voriconazole trough levels were not monitored during outpatient follow-up; however, he was closely observed for potential drug-related adverse effects. Serial brain CT evaluations were performed. Follow-up imaging at 3 months post-discharge demonstrated a reduction in the size of multiple residual cerebral abscesses. Oral voriconazole therapy was continued until complete radiologic resolution was achieved. He received oral voriconazole for a total of 12 months without any drug-related complications, and subsequent brain CT demonstrated complete resolution of the ring-enhancing lesions without midline shift. Currently, the patient is independent in activities of daily living and self-care, although he exhibits mildly slowed verbal responses. He has had no clinical features suggestive of recurrent infection following completion of therapy. DiscussionAspergillus is an opportunistic fungus commonly found in soil and decomposing vegetation. Among the Aspergillus species, A. fumigatus is the most frequently identified causative pathogen in cerebral aspergillosis, a rare but severe manifestation of invasive aspergillosis involving the central nervous system (CNS) infection with an extremely poor prognosis. Intracranial infections most commonly occur through hematogenous dissemination, whereas direct or contiguous spread from adjacent structures is less frequently observed (Marinovic et al., 2007). Clinical manifestations are often nonspecific and may include progressive headache, focal neurological deficits, fever, cranial nerve palsies, and seizures, which may contribute to delayed diagnosis (Miceli, 2019; Meena et al., 2021). A systematic review by Dotis et al. (2007) identified 90 cases of CNS aspergillosis in children under 18 years old, with brain abscesses representing the most common presentation and leukemia being the most frequent underlying condition. Ventriculitis, although less common, is often associated with particularly high fatality. The vulnerability of children receiving prolonged corticosteroid therapy was highlighted by Roilides E, et al. (2003), who reported a fatal case of pulmonary and cerebral aspergillosis in an 18-month-old boy with corticosteroid-resistant nephrotic syndrome, presenting with rapidly progressive coma and seizures. Corticosteroids impair host immune defenses by suppressing neutrophil recruitment, reducing monocyte and macrophage counts, and inhibiting both phagocytosis and the oxygen-independent fungicidal mechanisms, thereby significantly increasing susceptibility to invasive aspergillosis (Roilides et al., 2003). The VAV-ECMO circuit functions as a large foreign body that disrupts the normal skin barrier, while cannulas traversing blood vessels facilitate the formation of biofilm within the tubing and provide a surface on which fungal pathogens can proliferate. Furthermore, the warm, nutrient-rich blood flow within the circuit promotes pathogen growth and contributes to an increased infection burden. Moreover, the VAV-ECMO catheter and cannulas create multiple potential entry points for pathogens, increasing the overall risk of infection (Garcia et al., 2013). Patients requiring VAV- ECMO are typically critically ill and often receive prolonged courses of broad-spectrum antibiotics, which disrupt normal microbial flora. Concurrent corticosteroid therapy further compromises host immune responses by impairing neutrophil migration and function (Mustafa, 2023). In combination with VAV-ECMO therapy, the use of corticosteroids and prolonged broad-spectrum antibiotics are important predisposing factor for developing invasive fungal infections such as aspergillosis. Although fungal infections in VAV-ECMO patients are more commonly caused by Candida species (Bizzarro et al., 2011), A. fumigatus infections occur more frequently in high-risk populations, including patients with hematologic malignancies, prolonged corticosteroid use, immunosuppressive therapy, or solid organ transplantation, with reported incidence approaching nearly 100% in some series (Alessandri et al., 2025). Most reported cases of invasive aspergillosis primarily involve the lung parenchyma, with secondary dissemination to other organs (Garcia et al., 2013; Aubron et al., 2013; Parcell et al., 2014). In contrast, isolated cerebral aspergillosis without radiologic evidence of pulmonary or sinonasal disease, as observed in this case, is uncommon. Negative sputum and blood cultures do not identify fungal infection. Although the precise source of infection could not be identified, colonization of VAV-ECMO-related catheters or cannulas may have contributed to hematogenous seeding of the brain. Environmental exposure to airborne conidia is also a potential source; however, environmental sampling was not performed. These findings underscore the importance of strict infection-control measures, including routine maintenance of air-handling systems and meticulous care of intravascular devices in high-risk clinical settings. The diagnosis of cerebral aspergillosis remains challenging. Serum galactomannan testing has limited sensitivity in isolated CNS disease, and false-negative results have been reported, particularly in patients with localized infections or chronic granulomatous disease. Clinicians should therefore interpret negative results with caution. Definitive diagnosis relies on histopathological examination of the brain tissue, supported by fungal culture and molecular techniques such as polymerase chain reaction. Voriconazole, a triazole antifungal agent with potent activity against Aspergillus species and favorable CSF penetration, is currently recommended as first-line therapy for CNS aspergillosis. Routine primary combination antifungal therapy is not recommended; however, salvage or combination therapy may be considered in severe or refractory cases. Echinocandins used in combination with voriconazole or liposomal amphotericin B have demonstrated synergistic activity in selected cases (Patterson et al., 2016). In this patient, the limit of the used echinocandin and liposomal amphotericin B therapy. The patient was treated with voriconazole in combination with amphotericin B, as combination therapy has been reported to result in successful outcomes in selected adult patients with cerebral aspergillosis (Imbert et al., 2017). Despite aggressive antifungal treatment, clinical improvement was limited, and amphotericin B was discontinued after one month due to lack of symptomatic response. In addition to antifungal therapy, meropenem was administered to treat the multiple brain abscesses and to provide broad-spectrum coverage for potential aerobic and anaerobic pathogens. Broad-spectrum antimicrobial therapy was selected in accordance with current recommendations, with a treatment duration of 6–8 weeks to ensure adequate eradication of the infection (Bodilsen et al., 2024). Aggressive antifungal therapy combined with neurosurgical intervention is crucial for improving mortality and neurological outcomes in patients with cerebral aspergillosis. Standard management of fungal brain abscesses includes complete surgical excision in combination with antifungal therapy. However, in this case, the abscesses were multiple and located in deep or eloquent brain regions, rendering complete surgical removal high-risk. Consequently, total excision could not be achieved during the initial operation. Owing to the severity of the disease, extensive intracranial involvement, and recurrent increases in intracranial pressure requiring multiple neurosurgical interventions, prolonged voriconazole therapy was necessary and continued until radiologic resolution of the brain abscesses was achieved. This case highlights the importance of preventing invasive fungal infections through strict adherence to proper hand hygiene and aseptic techniques during catheter and VAV-ECMO cannula care. Early detection through routine microbial surveillance, including fungal cultures from all catheter and cannula sites, is essential for timely diagnosis. These measures may help improve outcomes in patients receiving VAV-ECMO support. Early diagnosis and a combined approach using systemic antifungal therapy and prompt neurosurgical intervention can be effective in managing multiple brain abscesses and improving outcomes for patients with refractory CNS aspergillosis (Elefanti et al., 2013; Patterson et al., 2016). ConclusionThis case underscores the interplay of multiple predisposing factors contributing to invasive fungal infections, particularly prolonged corticosteroid use and VAV-ECMO therapy, which may increase susceptibility to cerebral aspergillosis. Early recognition of this rare but life-threatening condition is essential, as timely diagnosis combined with aggressive systemic antifungal therapy and appropriate neurosurgical intervention may significantly improve survival and neurological outcomes. AcknowledgmentsThe authors would like to thank Ms. Daisy Jimenez Gonzales from the International Relations Section, Faculty of Medicine, Naresuan University, for her assistance with English language editing of the manuscript. Ethical approvalThis case report was approved by the Institutional Review Board, Faculty of Medicine, Naresuan University Hospital (IRB No. P3-0011/2567). Written informed consent from the legal guardian was obtained in accordance with ethical standards before publication. FundingNone. Authors’ contributionsChutima Phuaksaman, Jiroje Jiranukool, and Sarunya Srijuntongsiri contributed to the study concept and design. Sarunya Srijuntongsiri provided administrative support. Chutima Phuaksaman, Nuanluck Yupensuk, Veeratape Ngamnusonkit, Krittaporn Phruksarudee, and Sarunya Srijuntongsiri contributed to data collection and assembly. Chutima Phuaksaman, Jiroje Jiranukool, and Sarunya Srijuntongsiri performed data analysis and interpretation. All authors contributed to manuscript drafting and revision, and approved the final version of the manuscript. Conflicts of interestAll authors declare no conflicts of interest concerning this article’s research, authorship, and/or publication. Data availabilityAll data are provided in the manuscript. ReferencesAlessandri, F., Giordano, G., Sanda, V.C., D'Ettorre, G., Pugliese, F. and Ceccarelli, G. 2025. Outcomes of severe aspergillosis in patients undergoing extracorporeal membrane oxygenation: a systematic review. Artif. Organs 49(3), 362–372. Aubron, C., Pilcher, D., Leong, T., Cooper, D.J., Scheinkestel, C., Pellegrino, V. and Cheng, A.C. 2013. Aspergillus sp. isolated in critically ill patients with extracorporeal membrane oxygenation support. Scand. J. Infect. Dis. 45, 715–721. Bizzarro, M.J., Conrad, S.A., Kaufman, D.A. and Rycus, P. 2011. Infections acquired during extracorporeal membrane oxygenation in neonates, children, and adults. Pediatr. Crit. Care Med. 12, 277–281. Bodilsen, J., D'Alessandris, Q.G., Humphreys, H., Iro, M.A., Klein, M., Last, K., Montesinos, I.L., Pagliano, P., Sipahi, O.R., San-Juan, R., Tattevin, P., Thurnher, M., De J. Treviño-rangel, R. and Brouwer, M.C. 2024. European Society of Clinical Microbiology and Infectious Diseases guidelines on diagnosis and treatment of brain abscess in children and adults. Clin. Microbiol. Infect. 30(1), 66–89. Dotis, J., Iosifidis, E. and Roilides, E. 2007. Central nervous system aspergillosis in children: a systematic review of reported cases. Int. J. Infect. Dis. 11, 381–393. Elefanti, A., Mouton, J.W., Verweij, P.E., Tsakris, A., Zerva, L. and Meletiadis, J. 2013. Amphotericin B- and voriconazole-echinocandin combinations against Aspergillus spp.: effect of serum on inhibitory and fungicidal interactions. Antimicrob. Agents Chemother. 57, 4656–4663. Garcia, X., Mian, A., Mendiratta, P., Gupta, P., Rycus, P. and Prodhan, P. 2013. Aspergillus infection and extracorporeal membrane oxygenation support. J. Intensive Care Med. 28, 178–184. Imbert, S., Brossas, J.Y., Palous, M., Joly, I., Meyer, I. and Fekkar, A. 2017. Performance of Aspergillus PCR in cerebrospinal fluid for the diagnosis of cerebral aspergillosis. Clin. Microbiol. Infect. 23(11), 889.e1–889.e4. Marinovic, T., Skrlin, J., Vilendecic, M., Rotim, K. and Grahovac, G. 2007. Multiple Aspergillus brain abscesses in immunocompetent patient with severe cranio-facial trauma. Acta. Neurochir. (Wien). 149(6), 629–632. McCarthy, M., Rosengart, A., Schuetz, A.N., Kontoyiannis, D.P. and Walsh, T.J. 2014. Mold infections of the central nervous system. N. Engl. J. Med. 371, 150–160. Mccarthy, M.W., Kalasauskas, D., Petraitis, V., Petraitiene, R. and Walsh, T.J. 2017. Fungal Infections of the Central Nervous System in Children. J. Pediatric Infect. Dis. Soc. 6, e123–e133. Meena, D.S., Kumar, D., Bohra, G.K. and Kumar, G. 2021. Clinical manifestations, diagnosis, and treatment outcome of CNS aspergillosis: a systematic review of 235 cases. Infect. Dis. Now 51, 654–660. Miceli, M.H. 2019. Central nervous system infections due to aspergillus and other hyaline Molds. J. Fungi (Basel). 5, 79. Mustafa, S.S. 2023. Steroid-induced secondary immunodeficiency. Ann. Allergy. Asthma. Immunol. 130(6), 713–717. Parcell, B.J., B C Raju, P.K., Johnson, E.M., Fardon, T.C. and Olver, W.J. 2014. Invasive pulmonary aspergillosis post-extracorporeal membrane oxygenation support and literature review. Med. Mycol. Case Rep. 4, 12–15. Patterson, T.F., Thompson, G.R., Denning, D.W., Fishman, J.A., Hadley, S., Herbrecht, R., Kontoyiannis, D.P., Marr, K.A., Morrison, V.A., Nguyen, M.H., Segal, B.H., Steinbach, W.J., Stevens, D.A., Walsh, T.J., Wingard, J.R., Young, J.A.H. and Bennett, J.E. 2016. Practice Guidelines for the Diagnosis and Management of Aspergillosis: 2016 Update by the Infectious Diseases Society of America. Clin. Infect. Dis. 63, e1–e60. Roilides, E., Pavlidou, E., Papadopoulos, F., Panteliadis, C., Farmaki, E., Tamiolaki, M. and Sotiriou, J. 2003. Cerebral aspergillosis in an infant with corticosteroid-resistant nephrotic syndrome. Pediatr. Nephrol. 18, 450–453. Sullivan, B.N., Baggett, M.A., O'Connell, S.S., Pickett, K.M. and Steele, C. 2022. A systematic review to assess the relationship between disseminated cerebral aspergillosis, leukemias and lymphomas, and their respective therapeutics. J. Fungi. (Basel). 8, 722. | ||
| How to Cite this Article |
| Pubmed Style Srijuntonsiri S, Phuaksaman C, Phruksarudee K, Jiranukool J, Somran J, Yupensuk N, Ngamnusonkit V. Multiple cerebral abscesses caused by Aspergillus fumigatus following extracorporeal membrane oxygenation in a pediatric patient: A case report. doi:10.5455/JMID.2026.v16.i1.6 Web Style Srijuntonsiri S, Phuaksaman C, Phruksarudee K, Jiranukool J, Somran J, Yupensuk N, Ngamnusonkit V. Multiple cerebral abscesses caused by Aspergillus fumigatus following extracorporeal membrane oxygenation in a pediatric patient: A case report. https://www.jmidonline.org/?mno=292397 [Access: March 01, 2026]. doi:10.5455/JMID.2026.v16.i1.6 AMA (American Medical Association) Style Srijuntonsiri S, Phuaksaman C, Phruksarudee K, Jiranukool J, Somran J, Yupensuk N, Ngamnusonkit V. Multiple cerebral abscesses caused by Aspergillus fumigatus following extracorporeal membrane oxygenation in a pediatric patient: A case report. doi:10.5455/JMID.2026.v16.i1.6 Vancouver/ICMJE Style Srijuntonsiri S, Phuaksaman C, Phruksarudee K, Jiranukool J, Somran J, Yupensuk N, Ngamnusonkit V. Multiple cerebral abscesses caused by Aspergillus fumigatus following extracorporeal membrane oxygenation in a pediatric patient: A case report. doi:10.5455/JMID.2026.v16.i1.6 Harvard Style Srijuntonsiri, S., Phuaksaman, . C., Phruksarudee, . K., Jiranukool, . J., Somran, . J., Yupensuk, . N. & Ngamnusonkit, . V. (2026) Multiple cerebral abscesses caused by Aspergillus fumigatus following extracorporeal membrane oxygenation in a pediatric patient: A case report. doi:10.5455/JMID.2026.v16.i1.6 Turabian Style Srijuntonsiri, Sarunya, Chutima Phuaksaman, Krittaporn Phruksarudee, Jiroje Jiranukool, Julintorn Somran, Nuanluck Yupensuk, and Veeratape Ngamnusonkit. 2026. Multiple cerebral abscesses caused by Aspergillus fumigatus following extracorporeal membrane oxygenation in a pediatric patient: A case report. doi:10.5455/JMID.2026.v16.i1.6 Chicago Style Srijuntonsiri, Sarunya, Chutima Phuaksaman, Krittaporn Phruksarudee, Jiroje Jiranukool, Julintorn Somran, Nuanluck Yupensuk, and Veeratape Ngamnusonkit. "Multiple cerebral abscesses caused by Aspergillus fumigatus following extracorporeal membrane oxygenation in a pediatric patient: A case report." doi:10.5455/JMID.2026.v16.i1.6 MLA (The Modern Language Association) Style Srijuntonsiri, Sarunya, Chutima Phuaksaman, Krittaporn Phruksarudee, Jiroje Jiranukool, Julintorn Somran, Nuanluck Yupensuk, and Veeratape Ngamnusonkit. "Multiple cerebral abscesses caused by Aspergillus fumigatus following extracorporeal membrane oxygenation in a pediatric patient: A case report." doi:10.5455/JMID.2026.v16.i1.6 APA (American Psychological Association) Style Srijuntonsiri, S., Phuaksaman, . C., Phruksarudee, . K., Jiranukool, . J., Somran, . J., Yupensuk, . N. & Ngamnusonkit, . V. (2026) Multiple cerebral abscesses caused by Aspergillus fumigatus following extracorporeal membrane oxygenation in a pediatric patient: A case report. doi:10.5455/JMID.2026.v16.i1.6 |