| Research Article | ||
J. Microbiol. Infect. Dis., (2026), Vol. 16(1): 1–11 Research Article Cytotoxic, anti-coronavirus, and antioxidant activity of Hypericum triquetrifolium: Potential therapeutic applicationsRabia Alghazeer1*, Sana Elghmasi2, Mihayl Varbanov3, Stéphanie Philippot3, Samia Hawisa4, Maamar Sidati5, Bashir Lwaleed6, Salah Azwai7, Ibrahim Eldaghayes7, Fatim Gammoudi7, Aboubaker Garbaj8, Musa Alkasak9 and Ezdehar Althaluti101Chemistry Department, Faculty of Science, University of Tripoli, Tripoli, Libya 2Department of Biochemistry, Faculty of Medicine, University of Tripoli, Tripoli, Libya 3Laboratoire Lorrain de Chimie Moléculaire (L2CM), Université de Lorraine, Nancy, France 4Department of Histology and Genetics, Faculty of Medicine, University of Tripoli, Tripoli, Libya 5Department of Marine Biotechnology, Marine Biology Research Center, Tripoli, Libya 6School of Health Sciences, University of Southampton, Southampton, United Kingdom 7Department of Microbiology and Parasitology, Faculty of Veterinary Medicine, University of Tripoli, Tripoli, Libya 8Department of Food Hygiene and Control, Faculty of Veterinary Medicine, University of Tripoli, Tripoli, Libya 9Department of Pharmacy, Garabulli Higher Institute of Medical Sciences and Technology, Garabulli, Tripoli, Libya 10Department of Marine Chemistry and Physics, Marin Biology Research Center, Tripoli, Libya *Corresponding Author: Rabia Alghazeer. Chemistry Department, Faculty of Science, University of Tripoli, Tripoli, Libya. Email: rabia_alghazeer [at] yahoo.com Submitted: 14/09/2025 Revised: 01/12/2025 Accepted: 07/12/2025 Published: 01/01/2026 © 2026 Open Veterinary Journal
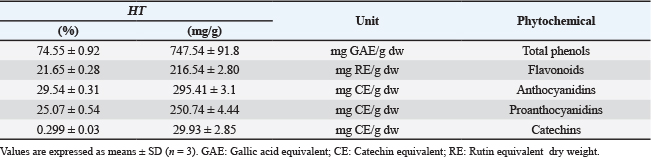
ABSTRACTBackground: Hypericum triquetrifolium (HT) is an herbal medicine that has promising pharmacological properties and is traditionally used to treat multiple diseases. Aim: This study examined the chemical composition, antioxidant, anti-inflammatory, and antiviral properties of HT, focusing on its impact on the cytopathic effect of human coronavirus-229E (HCoV-229E). Methods: The phytochemical content and in vitro bioactivity of the aqueous extract of HT collected from Al Marj, Libya, were documented. Common antioxidants were quantified, and the antioxidant activity of the extract was assessed using standard methods. The anti-inflammatory effect of albumin denaturation was evaluated. In this study, we determined the cytotoxicity on Medical Research Council-5 cells and antiviral activity by measuring the extract’s ability to inhibit the cytopathic effect of HCoV-229E on MRC-5 cells. Results: The phytochemical analysis confirmed the presence of phenols, flavonoids, proanthocyanidin, anthocyanidin, and catechin, and a high total phenolic content. The extract possessed strong free radical scavenging activity. In addition, the HT extract significantly inhibited heat-induced albumin denaturation, demonstrating its anti-inflammatory effect. The extract significantly inhibited the cytopathic effect of HCoV-229E on MRC-5 cells, indicating for the first time the antiviral effect of HT. Conclusion: To the best of our knowledge, this study is the first to demonstrate that HT extract has low cytotoxicity and potential antiviral activity against a surrogate human coronavirus. Although these results are promising, they are based on in vitro testing with HCoV-229E, and further studies, including in vivo and clinical investigations, are necessary to assess the relevance of HCoV-2 to severe acute respiratory syndrome-CoV-2 and other human coronaviruses. Keywords: Hypericum triquetrifolium, Flavonoids, Phenols, Antioxidant activity, COVID-19. IntroductionMany compounds from plants, or their metabolites, have promising medicinal potential, with several biological effects and a wide range of pharmacological properties, including immune system modulation, antimicrobial, anti-inflammatory, and antioxidant activities (Bilto et al., 2015; Al-Snafi, 2018; Menegazzi et al., 2020). Natural compounds are an essential source of novel drugs, as they are widely available and usually have low risks of side effects (Boukhatem and Setzer 2020). Among plants with traditional medicinal properties are species of the genus Hypericum, which belong to the Clusiaceae family and are found naturally in tropical and subtropical regions (Ghirga et al., 2021). Many Hypericum species have antioxidant, anti-depressive, and anti-cancer properties (Galati et al., 2008; Saddiqe et al., 2016; Menegazzi et al., 2021). Moreover, some of their phytochemicals exhibit potent antiviral activities (Al-Snafi, 2018). Therefore, they have been extensively investigated as a source of potential antivirals (Ismail, 2018; Akgöz, 2015). According to Yalçın et al. (2021), H. perforatum could be an effective antiviral against COVID-19 (Yalçın et al., 2021). Hypericum triquetrifolium (HT), which is closely related to H. perforatum, has attracted considerable scientific interest due to its anti-inflammatory, antioxidant, antimicrobial, and analgesic activities (Barış et al., 2011; Rouis et al., 2013; Saad et al., 2016). Hypericum triquetrifolium is a perennial herbaceous plant belonging to the family Clusiaceae. It typically grows to 30–70 cm in height, with triangular stems, opposite leaves, and bright yellow flowers measuring 1–2 cm in diameter. The species is naturally distributed in the Mediterranean and subtropical regions and is traditionally used for its anti-inflammatory, antimicrobial, and antioxidant properties. Human viral infections, such as Middle East Respiratory Syndrome, severe acute respiratory syndrome (SARS-CoV), and H1N1 influenza, pose major global health issues, especially those with high morbidity and mortality (Das et al., 2016; Lombardi et al., 2020; Petersen et al., 2020). In late 2019, an epidemic of lower respiratory tract infections with a high mortality rate emerged in Wuhan, China, and spread worldwide (Lu et al., 2020). COVID-19 is attributed to the novel SARS-CoV-2 virus belonging to the coronavirus family (Sharma et al., 2020). However, many uncertainties remain regarding the interaction of this virus with the host, transmission, clinical spectrum, diagnostics, prevention, and therapeutic strategies (Cascella et al., 2020; Li et al., 2023). To date, the clinical management of COVID-19 patients is based on symptomatic treatment, which highlights the need for novel therapeutic strategies (Chen et al., 2021). This study assessed the potential therapeutic effect of Libyan-grown HT extract, including its cytotoxic, antioxidant, and antiviral properties, particularly on SARS-CoV-2 surrogate viruses, such as HCoV 229E. Although the antiviral potential of certain Hypericum species has been documented, the biological activity of H. triquetrifolium, particularly against coronaviruses, is lacking. Moreover, no studies have evaluated the cytotoxicity, antioxidant capacity, or antiviral efficacy of extracts of Libyan-grown HT. This gap underscores the need to investigate HT as a potential natural therapeutic candidate with multi-target properties. Therefore, this study aimed to evaluate the cytotoxic, antioxidant, and antiviral activities of HT extract, focusing on its effects against the human coronavirus HCoV-229E as a SARS-CoV-2 surrogate. Materials and MethodsPlant materialFresh HT leaves were collected in May 2021 from Al Marj, Libya (Latitude: 32° 29' 16.73" N Longitude: 20° 49' 54.26" E). Mohamed Nuri Abuhadra, Professor of Plant Taxonomy, Department of Botany, Faculty of Science, University of Tripoli, Tripoli-Libya, authenticated them (Voucher number, 68211). The leaves were cleaned, dried, ground, and stored at 4°C. Cells and mediaThe human lung fibroblast cell line (MRC-5, ECACC 05090501, European Collection of Authenticated Cell Cultures, Salisbury, England) was grown in antibiotic-free minimum essential medium (Minimum Essential Medium, Sigma-Aldrich, France) supplemented with 10% fetal bovine serum (FBS) (Eurobio, France). MEM with 2% FBS was used for viral infection and the antiviral assays. VirusThe HCoV 229E strain (ATCC VR 740, USA) was titrated in MRC-5 cells. Cells (104cells/well) were cultured in 96-well plates in the presence of HCoV-229E for 72 hours at 33°C under a 5% CO2 atmosphere with a 10-fold serially diluted virus suspension. All virus stocks were stored at −80°C until further use. Extract preparationExtracts were prepared by shaking HT powder (5 g) in deionized water (200 ml) for 2 hours at 90°C. The samples were filtered using the Whatman filter paper No. 1, and the filtrate was freeze-dried in a freeze dryer (Gefriertrocknungsanlagen GmbH, Germany). The water extraction yield was 10.6%. Phytochemicals quantificationThe total phenolic content was quantified using the Folin–Ciocalteu method, as described by Viuda-Martos et al. (2011) and expressed as mg of gallic acid equivalent per gram of dry weight (mg GAE/g dw). The total flavonoid content was determined using a colorimetric assay with rutin as a standard, following the procedure described by Singleton et al. (1999). Results were expressed as mg of rutin equivalent per gram of dry weight (mg RE/g dw). The total proanthocyanidin concentration was determined as described by Schofield et al. (2001), expressed as mg catechin equivalent per gram of dry weight (mg CE/g dw). The total anthocyanidin concentration was determined using the vanillin-hydrochloric acid colorimetric method (Schofield et al., 2001), expressed as mg CE/g dw. The total catechin content was determined following the protocol of Nakamura et al. (2003), expressed as mg CE/g dw. Determination of antioxidant activitiesThe free radical scavenging activity of the HT extract (6.25–100 µg/ml) was determined using in vitro DPPH and ABTS antioxidant assays, as previously described (Re et al., 1999; Wong et al., 2006). Ascorbic acid (AA) and butylated hydroxyanisole (BHA) were used as positive controls. Determination of radical scavenging activity of hydroxylThe hydroxyl radical scavenging activity was assessed by quantifying the 2-deoxy-d-ribose degradation product, which forms a pink chromogen upon heating with thiobarbituric acid at low pH (Ilavarasan et al., 2005). Ascorbic acid was used as a standard (6.25–100 µg/ml). Determination of lipid peroxidation activityThe lipid peroxidation activity was measured as described by Kikuzaki and Nakatani (1993). Ascorbic acid was used as a standard 6.25–100 µg/ml). For all assays, the percentage of radical scavenging activity and lipid peroxidation inhibition was calculated using the following equation: (%)=[(AC—AS)/AC] × 100 where AC represents the absorbance of the control sample, and AS is the absorbance of the sample with the extract or standard. In vitro anti-inflammatory activityThe anti-inflammatory activity of the HT extract at 6.25–100 µg/ml was determined using the method described by Williams et al. (2008), which relies on albumin denaturation inhibition. BHA and AA served as positive controls. The percentage inhibition of albumin was calculated as follows: % inhibition of denaturation=(1 − D/C) ×100% where D and C are the absorbances of the test and control samples, respectively. Cytotoxicity assayMRC-5 cells were seeded in a 96-well plate (1.5 × 10 (Barış et al., 2011) cells/well) and incubated at 37°C under a 5% CO2 atmosphere. The dry HT extract was dissolved in dimethylsulfoxide (Biosolve, France) or sterile water and further diluted in MEM containing 2% FBS. Next, 24 hours after the initial seeding, the cells were treated with 100 µl of serially diluted HT extracts (3.12–1,000 µg/ml) and further incubated for 72 hours under identical conditions. Cell viability was determined using the MTT assay (Mosdam, 1983). The CC50 of the extract, corresponding to the concentration needed to reduce the MTT signal by 50%, was calculated. The survival percentage of extract-treated cells relative to control cells was calculated, and the maximum extract concentration showing no cytotoxic effect was determined. Antiviral testsThe antiviral activity of the HT extract was assessed by measuring the cytopathogenic effect of HCoV-229E on MRC-5 cells. The cells were seeded (1.5 × 10 (Barış et al., 2011) cells/well) in a 96-well plate 24 hours before virus inoculation. On the following day, the culture medium was removed, and a mixture consisting of 50 µg/ml or 100 µg/ml HT extract (based on MTT assay data) and a 1:10 serially diluted HCoV 229E virus suspension at multiplicity of infection (MOIs) of 0, 4, 0.04, and 0.004 was added to the cells. The cells were further incubated under identical conditions and examined for virus-induced cytopathic effect (CPE) 1, 2, and 3 days after virus inoculation under an optical microscope. CPE was determined when cell damage in untreated cultures was at its maximum. The TCID50 was determined, titrations were performed in triplicate, and titers were calculated in TCID50/ml. The CPE was estimated using the crystal violet uptake assay (Feoktistova et al., 2016). Virus-induced CPE and morphological changes in cells were observed using an optical microscope. Three independent experiments were performed in triplicate, and imaging and image processing were performed using an inverted fluorescence microscope (Zeiss, France). Statistical analysisStatistical analysis was performed using SPSS (Statistical Program for Social Sciences) v.16 (SPSS, Chicago, IL, USA). Data are presented as the mean ± standard deviation (SD). Significance was assessed using one-way ANOVA followed by Tukey’s multiple comparisons test with a significance level set at p ≤ 0.05. For cytotoxicity and antiviral studies, a t-test analysis was performed using GraphPad Prism 5 software. Ethical approvalNot needed for this study, as this article does not contain any studies conducted by any of the authors involving human or animal participants. ResultsQuantitative analysis of phytochemicalsTable 1 summarizes the phytochemical content of HT and details the levels of phenols, flavonoids, anthocyanins, anthocyanidins, proanthocyanidins, and catechins. The total phenolic compounds were more abundant than the other quantified bioactive compounds. Table 1. Total phenols, flavonoids, anthocyanins, anthocyanidins, proanthocyanidins, and catechins in aqueous HT extract.
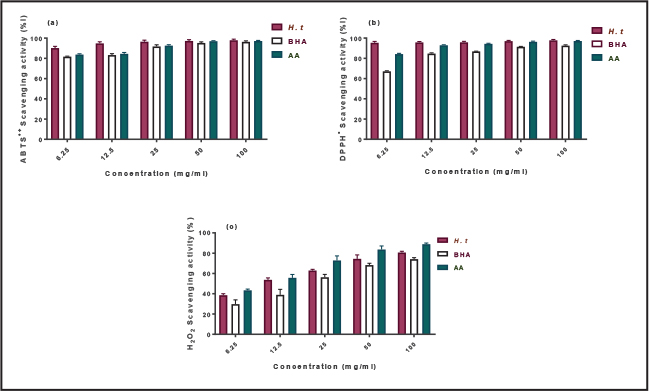
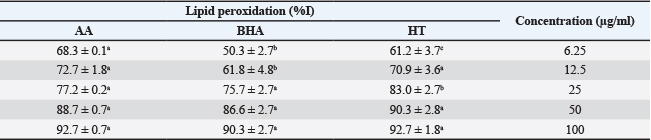
Antioxidant activityRadical scavenging capacity Radical scavenging capacity was measured using 2,2-diphenyl-1-picrylhydrazyl (DPPH) and 2,2′-azinobis (Arreola et al., 2015)-ethylbenzothiazoline-6-sulfonic acid (ABTS) assays and expressed as the percent inhibition of radical activity (Fig. 1). The ability of the HT extract to scavenge ABTS and DPPH radicals was compared with that of the standards BHA and AA.
Figure 1. Scavenging activity of the aqueous leaf extract of HT and positive controls against: (A) ABTS- radicals, (B) DPPH, and (C) hydroxyl radicals. Data are presented as mean ± SD (n=3). Bars labeled with different lowercase letters are significantly different (p < 0.05). AA: ascorbic acid; BHA: butylated hydroxyanisole. At low concentrations, the ABTS scavenging activity of the extract was significantly higher than that of the standards (p < 0.05). However, no noticeable differences were observed at high concentrations (Fig. 1A and B) shows the DPPH radical scavenging activity of the standards and aqueous HT extract at different concentrations. At a low concentration (6.25 µg/ml), the aqueous HT extract displayed higher radical scavenging activity than the standards (p < 0.05). At this concentration, the scavenging effects were 93.3%, 84.1%, and 67.1% for the HT extract, BHA, and ascorbic acid, respectively. The scavenging activity of all compounds was concentration-dependent, reaching a maximum at 100 µg/ml. No statistically significant differences were observed at this concentration. Hydroxyl radical scavenging activityThe effect of the HT extract on the formation of the hydroxyl radical (•OH) was evaluated. The HT extract exhibited a potent concentration-dependent hydrogen peroxide scavenging capacity (Fig. 1C). From 6.25 to 100 µg/ml, the H2O2 scavenging capacity ranged from 38.5% to 80.6%. At 100 µg/l, the HT extract inhibited 80.6% of the hydroxyl radical formation, which was significantly higher than that of BHA (74.0%, p < 0.05) but markedly lower than that of ascorbic acid (88.8%) at the same concentration. Lipid peroxidation inhibitionTable 2 shows the inhibitory action of the HT extract on hemoglobin-induced lipid peroxidation in the linoleic acid system. All the tested compounds reduced lipid peroxidation in a dose-dependent manner. At 6.25 µg/ml, the HT extract inhibited lipid peroxidation significantly more than both ascorbic acid and BHA (p < 0.05). However, no significant differences were observed between the compounds at 50 µg/ml and 100 mg/ml (p > 0.05). Table 2. Lipid peroxidation oxidation by the aqueous extract of the Hypericum triquetraifolium (HT) leaf, ascorbic acid (AA), and butylated hydroxyanisole (BHA).
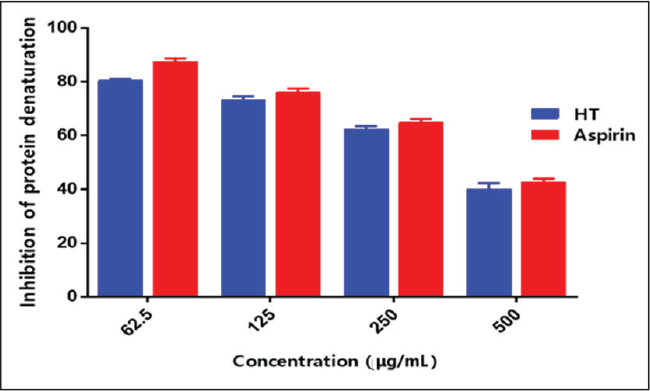
Anti-inflammatory activityBecause inflammation is correlated with protein denaturation, the anti-inflammatory activity of the HT extract was evaluated by quantifying the denaturation of egg albumin. The HT extract inhibited protein/albumin denaturation in a concentration-dependent manner (Fig. 2). The highest inhibition (86%) was observed at 1000 μg/ml. However, the HT extract exhibited lower inhibition rates than the reference drug.
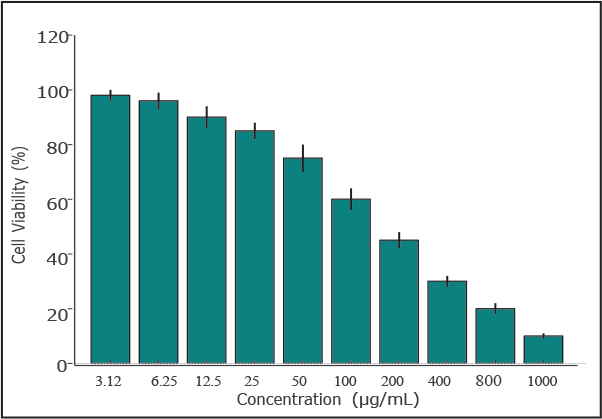
Figure 2. Inhibitory effect of the aqueous leaf extract of HT and aspirin (positive control) on heat-induced protein denaturation. Results are expressed as mean ± standard deviation (n=3). CytotoxicityThe cytotoxicity of the HT extract was assessed in MRC-5 cells. The CC50 (50% cytotoxicity concentration) values were around 500 µg/ml (Fig. 3A). At 50 and 100 µg/ml, the extract exhibited mild or no cytotoxicity. These two concentrations were used for the antiviral tests (Fig. 3B).
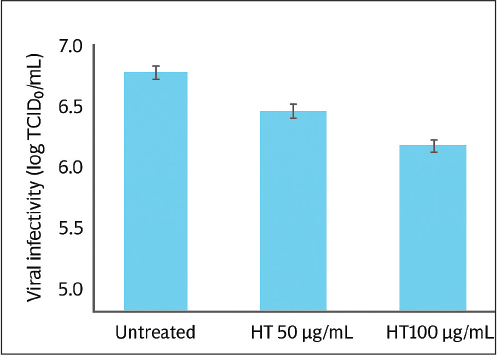
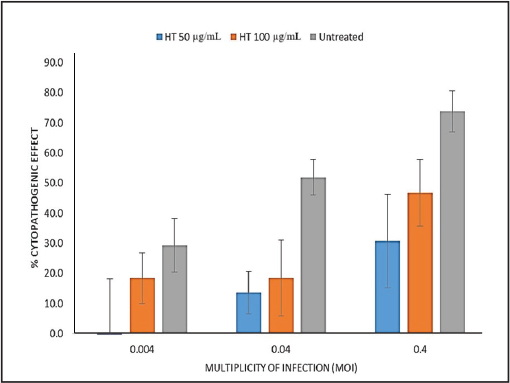
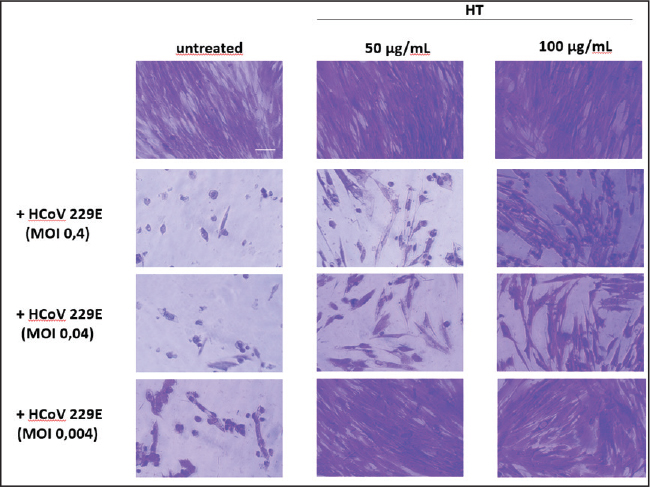
Figure 3. Cytotoxicity of Hypericum triquetrifolium (HT) leaf extract in MRC-5 human lung fibroblast cells following treatment with concentrations ranging from 3.12 to 1,000 µg/ml. Data are presented as mean ± standard deviation (n=3). Antiviral activityVirus-induced CPE occurred 72 hours after infection. The virus was titrated, and the median tissue culture infectious dose (TCID50)/ml in extract-treated and control samples was determined. After 72 hours, the HT extract reduced the viral titer by >1 log10 and 1.5 log10 at 50 and 100 µg/ml, respectively (Fig. 4). The antiviral effect of the extract was assessed by determining the reduction in virus-induced CPE using monolayers of cells stained with crystal violet. Without the HT extract, HCoV 229E had a lower CPE at low MOI (Fig. 5). No CPE was observed in infected cells with an MOI of 0.004 treated with 50 µg/ml HT extract. At 100 µg/ml, the extract reduced CPE by more than 10%. The CPE reduction remained significant at higher MOIs; at 50 µg/ml, the extract reduced CPE by 38% and 43% at MOIs of 0.04 and 0.4, respectively. The 100 µg/ml dose of extract yielded a more moderate antiviral effect and reduced the CPE by 33.4% and 27% at MOIs of 0.04 and 0.4, respectively. This reduced effect is probably due to the slightly higher cytotoxicity of the extract at this concentration. Our observations under a microscope confirmed the substantial reduction of HCoV 229E-induced CPE (Fig. 6), particularly at lower MOI (0.004), independent of the extract concentration.
Figure 4. Antiviral activity of HT leaf extract against human coronavirus 229E (HCoV−229E). Viral infectivity, expressed as log TCID50/ml, is shown for cells treated with different concentrations of HT extract (50 µg/ml and 100 µg/ml) compared with the untreated control. Data represent mean ± standard deviation (SD) (n=3).
Figure 5. Cytopathogenic Effect of Human Coronavirus 229E (HCoV−229E) on MRC-5 Cells Treated with Hypericum triquetrifolium (HT) Extract 72 hours post-infection, data are presented as mean ± standard deviation (n=3).
Figure 6. Cytopathogenic effect of human coronavirus 229E (HCoV−229E) on MRC-5 Cells in the Presence or Absence of Hypericum triquetrifolium (HT) Extract 72 Hours Post-Infection. Cells were fixed and stained with crystal violet and then imaged using bright-field optical microscopy. Scale bar=100 µm. DiscussionBioactive molecules of natural origin, such as phytochemicals, are a major source of novel drugs. They can exhibit potent antioxidant, anti-inflammatory, and antiviral activities (Al-Snafi, 2018; Yahfoufi et al., 2021). Therefore, plants are the subject of extensive investigation as a source of potential therapeutic agents. This study aimed to document the cytotoxic, antioxidant, and antiviral properties of the HT extract. In particular, we investigated whether the HT extract could protect against the CPE of the HCoV 229E strain using MRC-5 cells. The data presented here show that Libyan HT contains phenolics, flavonoids, anthocyanins, anthocyanidins, proanthocyanidins, and catechins with a significantly higher amount of total phenolics than other quantified bioactive compounds. The high phenolic content of the HT extract explains its biological properties, as this class of compounds is known to be a major contributor to plant extracts’ antioxidant and anti-inflammatory activities of plant extracts (Fraga et al., 2019; Gulcin, 2020). The reducing property of a compound is generally attributed to its reductant content, such as phenolics and flavonoids. The antioxidant mechanism of these natural compounds primarily arises from their ability to donate hydrogen atoms or electrons, thereby neutralizing free radicals and preventing various oxidative chain reactions (Yahfoufi et al., 2018; Gulcin, 2020). According to this principle, the aqueous HT extract exhibited a notable, concentration-dependent hydrogen peroxide scavenging capacity. Therefore, HT can be considered a promising source of natural antioxidants. The aqueous extracts of HT exhibited potent radical scavenging activity; at 6.25 µg/ml, its efficacy was markedly higher than that of standard antioxidants at 6.25 g/ml. This high reducing power can be attributed to the strong electron-donating capacity of the phenolic constituents of the extract, as evidenced by its ability to prevent deoxyribose degradation primarily through direct hydroxyl radical neutralization rather than metal chelation (a mechanism consistent with that described by Barış et al., 2011). Furthermore, the extract significantly inhibited lipid peroxidation, underscoring its potential to protect biological systems from oxidative damage (Lorenzo et al., 2018). Previous studies have substantiated the significance of the observed antioxidant activity of HT (Conforti et al., 2002; Conforti et al., 2007; Kmail, 2022). In addition, the major role of antioxidants against different diseases underscores the significance of the observed antioxidant activity of the HT extract (Maxwell and Lip, 1997; Tandon et al., 2005; Rani, 2017; Pary and Markad, 2023). HT and other herbal medicines have been widely studied for their anti-inflammatory properties. However, few studies have evaluated their effect on inflammation. Therefore, the anti-inflammatory properties of HT were investigated, which revealed that HT extracts effectively inhibited protein/albumin denaturation, indicating its anti-inflammatory activity. Saad et al. (2011) suggested that the anti-inflammatory effect of HT stems from its ability to suppress the expression of inducible nitric oxide synthase and tumor necrosis factor alpha expressions. In addition, the immune system releases pro-inflammatory cytokines as an initial response to infection or irritation. Cytokine release inhibitors are good anti-inflammatory drug candidates (Arreola et al., 2015). Next, we examined the cytotoxic effect of HT extracts and found low or no cytotoxic effect on the human lung fibroblast cell line (MRC-5), cells at 50 and 100 µg/ml. Such low toxicity is another promising aspect of this plant as a candidate for new therapeutic approaches. This study is the first to document the antiviral activity of HT extracts and support a possible role of HT against human coronaviruses by demonstrating that the HT extract inhibits the cytopathic activity of HCoV 229E. Since several molecules found in plant cells are lipophilic and can thus penetrate membranes, the viral envelope may be the site of action (Zeedan and Abdalhamed, 2023). The molecules present in the extract may also affect the ability of the virus to bind to the host cell, preventing viral invasion. Furthermore, the viral envelope glycoproteins may be affected equally by the HT extract. The direct antiviral effect indicated by the CPE reduction, along with the demonstrated antioxidant and anti-inflammatory properties, further support the potential role of HT compounds in managing the pathogenic consequences of coronavirus infection (Mani et al., 2020; Zaidi and Taher, 2021). These compounds could help reduce the morbidity and mortality associated with COVID-19 by targeting the virus itself, regulating the immune response, and suppressing damaging inflammatory reactions, such as cytokine release syndrome (Kumar et al., 2021; Costa et al., 2023). Further studies are needed to clarify the exact anti-inflammatory mechanism of this plant extract and to identify the specific constituent responsible for this activity. ConclusionThis study offers preliminary in vitro evidence that HT extract is a promising, low-cytotoxicity, multi-target therapeutic candidate. Its bioactive compounds show potential against viral infections, specifically a surrogate coronavirus (HCoV-229E), suggesting synergistic antiviral, anti-inflammatory, and immunomodulatory effects. However, these findings are based solely on in vitro tests, meaning that the efficacy and safety of the extract are currently unproven. Its relevance to SARS-CoV-2 and other human coronaviruses needs to be confirmed in vivo, especially considering potential issues such as low human bioavailability and metabolic variability. The precise mechanism of action is unclear, complicating the prediction of drug interactions and long-term safety with prolonged use. Therefore, these results are exploratory and provide a foundation for future research rather than justifying clinical use. Future work must begin with animal studies to characterize pharmacokinetics and toxicity, followed by phased clinical trials to assess safety, optimal dosing, and therapeutic efficacy. Ultimately, standardizing the extract and elucidating its molecular targets are crucial steps for translating this finding into a viable therapeutic option. AcknowledgmentsNone. Conflict of interestThe authors have no conflicts of interest to declare. FundingThis study received no external funding. Authors' contributionsAll authors contributed to reading and approving the manuscript. Data availabilityThe original contributions presented in this study are included in the article/Supplementary material, and further inquiries can be directed to the corresponding author. ReferencesAkgöz, J. 2015. The effects of Hypericum (Hypericaceae) species on microorganisms. A review. Int. Res. J. Pharm. 6(7), 390–399. Al-Snafi, A.E. 2018. Chemical constituents and pharmacological effects of Hypericum triquetrifolium. Indian J. Pharm. Sci. 5(3), 1757–1765. Arreola R, Quintero-Fabián S, López-Roa RI, Flores-Gutiérrez EO, Reyes-Grajeda JP, Carrera-Quintanar L, Ortuño-Sahagún D. 2015. Immunomodulation and anti-inflammatory effects of garlic compounds. J Immunol Res. 401630. doi: 10.1155/2015/401630. Barış, D., Kızıl, M., Aytekin, C., Kızıl, G., Yavuz, M., Çeken, B. and Ertekin, A.S. 2011. In vitro antimicrobial and antioxidant activity of ethanol extract of three Hypericum and three Achillea species from Turkey. Int. J. Food. Prop. 14(2), 339–355. Bilto, Y., Alabdallat, N. and Salim, M. 2015. Antioxidant properties of twelve selected medicinal plants commonly used in Jordan. Br. J. Pharm. Res. 6(2), 121–130. Boukhatem, M.N. and Setzer, W.N. 2020. Aromatic herbs, medicinal plant-derived essential oils, and phytochemical extracts as potential therapies for coronaviruses: future perspectives. Plants 9(6), 800. Cascella, M., Rajnik, M., Cuomo, A., Dulebohn, S.C. and Di Napoli, R. 2020. Features, Evaluation and Treatment Coronavirus (COVID-19). https://www.ncbi.nlm.nih.gov/books/NBK554776 Chen, B., Liu, M. and Huang, C. 2021. Current diagnostic and therapeutic strategies for COVID-19. J. Pharm. Anal. 11(2), 129–137. Conforti, F., Statti, G.A., Tundis, R., Menichini, F. and Houghton, P. 2002. Antioxidant activity of methanolic extract of Hypericum triquetrifolium Turra aerial part. Fitoterapia 73(6), 479–483. Costa, M.F., Durço, A.O., Rabelo, T.K., Barreto, R.S.S. and Guimarães, A.G. 2023. Effects of Carvacrol, Thymol and essential oils containing such phenols on inflammation and the immune system: a comprehensive review of preclinical and clinical evidence. Front. Pharm. 14, 1134870. Feoktistova, M., Geserick, P. and Leverkus, M. 2016. Crystal Violet Assay for Determining Viability of Cultured Cells. Cold. Spring. Harb. Protoc. 1(4), 343–6. doi: 10.1101/pdb.prot087379 Fraga, C.G., Croft, K.D., Kennedy, D.O. and Tomás-Barberán, F.A. 2019. The effects of polyphenols and other bioactives on human health. Brit. J. Nutrit. 121(8), 863–865. Galati, E.M., Contartese, G., Miceli, N., Taviano, M.F., Sdrafkakis, V., Couladis, M. and Lanuzza, F. 2008. Antiinflammatory and antioxidant activity of Hypericum rumeliacum Boiss. subsp. apollinis (Boiss. & Heldr.) Robson & Strid methanol extract. Phytotherapy Res. Int. J. Devoted To Pharmacol. Toxicological Eval. Natural Product Derivatives 22(6), 766–771. Ghirga, F., Quaglio, D., Mori, M., Cammarone, S., Iazzetti, A., Goggiamani, A., Ingallina, C., Botta, B. and Calcaterra, A. 2021. A unique high-diversity natural product collection as a reservoir of new therapeutic leads. Org. Chem. Front. 8(5), 996–1025. Gulcin. 2020. Antioxidants and antioxidant methods: an updated overview. Arch. Toxicol. 94(3), 651–715. Ilavarasan, R., Malika, M. and Venkataraman, S. 2005. Anti-inflammatory and antioxidant activities of Cassia fistula Linn bark extracts. Afr. J. Tradit. Complement. Altern. Med. 2(1), 70–85. Ismail, F. 2018. Boosting hypericin and herb yield of Hypericum triquetrifolium L. by leaf enlargement. Adv. Environ. Biol. 12(1), 11–16. Kikuzaki, H. and Nakatani, N. 1993. Antioxidant effects of some ginger constituents. J. Food. Sci. 58(6), 1407–1410. Kmail, A. 2022. Protective Role of Hypericum perforatum L. and Hypericum triquetrifolium Turra against Inflammatory Diseases: evidence from in vitro and in vivo studies. Eur. J. Med. Plants 33(12), 34–47. Kumar, S., Sharma, P.P., Upadhyay, C., Kempaiah, P. and Rathi, B. 2021. Harnessing plant biodiversity for the discovery of multi-targeted phytochemicals against COVID-19. South. Afr. J. Botany. 143, 158–169. Li, G., Hilgenfeld, R., Whitley, R. and De Clercq, E. 2023. Therapeutic strategies for COVID-19: progress and lessons learned. Nat. Rev. Drug. Discov. 22(6), 449–475. Lombardi, A.F., Afsahi, A.M., Gupta, A. and Gholamrezanezhad, A. 2021. Severe acute respiratory syndrome (SARS), Middle East respiratory syndrome (MERS), influenza, and COVID-19, beyond the lungs: a review article. Radiol. Med. 126(4), 561–569. Lorenzo, J.M., Munekata, P.E., Sant'Ana, A.S., Carvalho, R.B., Barba, F.J., Toldrá, F. and Mora, L. 2018. Main characteristics of peanut skin and its role for the preservation of meat products. Trends Food Sci. Technol. 77, 1–10. Lu, H., Stratton, C.W. and Tang, Y.W. 2020. Outbreak of pneumonia of unknown etiology in Wuhan, China: the mystery and the miracle. J. Med. Virol. 92(4), 401. Mani, J.S., Johnson, J.B., Steel, J.C., Broszczak, D.A., Neilsen, P.M., Walsh, K.B. and Naiker, M. 2020. Natural product-derived phytochemicals as potential agents against coronaviruses: a review. Virus. Res. 284, 197989. Maxwell, S.R. and Lip, G.Y. 1997. Free radicals and antioxidants in cardiovascular disease. Br. J. Clin. Pharmacol. 44(4), 307–317. Menegazzi, M., Masiello, P. and Novelli, M. 2020. Anti-tumor activity of Hypericum perforatum L. and hyperforin through modulation of inflammatory signaling, ROS generation and proton dynamics. Antioxidants 10(1), 18. Mosdam, T.J.J.I.M. 1983. Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxic assay. J. Immunol. Methods 65, 55–63. Nakamura, Y., Tsuji, S. and Tonogai, Y. 2003. Analysis of proanthocyanidins in grape seed extracts, health foods and grape seed oils. J. Health Sci. 49(1), 45–54. Rani. 2017. Role of antioxidants in prevention of diseases. J. Appl. Biotechnol. Bioeng. 4(1), 91. Re, R., Pellegrini, N., Proteggente, A., Pannala, A., Yang, M. and Rice-Evans, C. 1999. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free. Radic. Biol. Med. 26(9-10), 1231–1237. Rouis, Z., Abid, N., Koudja, S., Yangui, T., Elaissi, A., Cioni, P.L., Flamini, G. and Aouni, M. 2013. Evaluation of the cytotoxic effect and antibacterial, antifungal, and antiviral activities of Hypericum triquetrifolium Turra essential oils from Tunisia. BMC. Complement. Altern. Med. 13, 24; doi:10.1186/1472-6882-13-24 Saad, B., AbouAtta, B. S., Basha, W., Hmade, A., Kmail, A., Khasib, S., & Said, O. 2011. Hypericum triquetrifolium-Derived Factors Downregulate the Production Levels of LPS-Induced Nitric Oxide and Tumor Necrosis Factor-α in THP-1 Cells. Evid. Based Complement. Alternat. Med. 2011(1), 586470. Saad, B., Embaslat, W. H., Abu-Farich, B., Mahajna, S., & Azab, M. 2016. Hypericum triquetrifolium extracts modulate IL-6, IL-10 and TNF-α protein and mRNA expression in LPS-activated human peripheral blood mononuclear cells and THP-1-derived macrophages. Med. Aromat. Plants S, 3, 2167-0412. Saddiqe, Z., Maimoona, A., Abbas, G., Naeem, I. and Shahzad, M. 2016. Pharmacological screening of Hypericum androsaemum extracts for antioxidant, anti-lipid peroxidation, antiglycation and cytotoxicity activity. Pak. J. Pharm. Sci. 29(2), 397–405. Schofield, P., Mbugua, D.M. and Pell, A.N. 2001. Analysis of condensed tannins: a review. Anim. Feed. Sci. Technol. 91(1-2), 21–40. Sharma, A., Tiwari, S., Deb, M.K. and Marty, J.L. 2020. Severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2): a global pandemic and treatment strategies. Int. J. Antimicrob. Agents. 56(2), 106054. Singleton, V.L., Orthofer, R. and Lamuela-Raventós, R.M. 1999. Analysis of total phenols and other oxidation substrates and antioxidants by means of Folin-Ciocalteu reagent. Methods. Enzymol. 1999, 152–178. Standl, F., Jöckel, K.H., Brune, B., Schmidt, B. and Stang, A. 2020. Comparing SARS-CoV-2 with SARS-CoV and influenza pandemics. Lancet. Infect. Dis. 20(9), e238–e244; doi:10.1016/S1473-3099(20)30484-9 Tandon, R., Khanna, H.D. and Dorababu, M. 2005. Oxidative stress and antioxidants in health and disease. Med. J. Armed Forces India 61(3), 258–63. Viuda-Martos, M., Ruiz-Navajas, Y., Fernández-López, J., Sendra, E., Sayas-Barberá, E. and Pérez-Álvarez, J.A. 2011. Antioxidant properties of pomegranate (Punica granatum L.) bagasses obtained as co-product in the juice extraction. Int. Food. Res. J. 44(5), 1217–1223. Williams, L.A., O'Connar, A., Latore, L., Dennis, O., Ringer, S., Whittaker, J.A., Conrad, J., Vogler, B., Rosner, H. and Kraus, W. 2008. The in vitro anti-denaturation effects induced by natural products and non-steroidal compounds in heat treated (immunogenic) bovine serum albumin is proposed as a screening assay for the detection of anti-inflammatory compounds, without the use of animals, in the early stages of the drug discovery process. West Indian Med. J. 57(4), 327–331. Wong, C.C., Li, H.B., Cheng, K.W. and Chen, F. 2006. A systematic survey of antioxidant activity of 30 Chinese medicinal plants using the ferric reducing antioxidant power assay. Food. Chem. 97(4), 705–711. Yahfoufi, N., Alsadi, N., Jambi, M. and Matar, C. 2021. The immunomodulatory and anti-inflammatory role of polyphenols. Nutrients 10(11), 1618. Yalçın, S., Yalçınkaya, S. and Ercan, F. 2021. Determination of potential drug candidate molecules of the Hypericum perforatum for COVID-19 treatment. Curr. Pharmacol. Rep. 7(2), 42–48. Zaidi, S.F.H. and Taher, M. 2021. Natural products and their bioactive compounds: a promising strategy for the treatment of COVID-19. Saudi J. Biol. Sci. 28(1), 865–875. Zeedan, G. and Abdalhamed, A. 2023. Antiviral Plant Extracts: a Treasure for Treating Viral Diseases. IntechOpen, London, ; doi:10.5772/intechopen.111732 | ||
| How to Cite this Article |
| Pubmed Style Alghazeer R, Elghmasi S, Varbanov M, Philippot S, Hawisa S, Sidati M, Lwaleed B, Azwai S, Eldaghayes I, Gammoudi F, Garbaj A, Alkasak M, Althaluti E. Cytotoxic, anti-coronavirus, and antioxidant activity of Hypericum triquetrifolium: Potential therapeutic applications. J Microbiol Infect Dis. 2026; 16(1): 1-11. doi:10.5455/JMID.2026.v16.i1.1 Web Style Alghazeer R, Elghmasi S, Varbanov M, Philippot S, Hawisa S, Sidati M, Lwaleed B, Azwai S, Eldaghayes I, Gammoudi F, Garbaj A, Alkasak M, Althaluti E. Cytotoxic, anti-coronavirus, and antioxidant activity of Hypericum triquetrifolium: Potential therapeutic applications. https://www.jmidonline.org/?mno=283728 [Access: January 25, 2026]. doi:10.5455/JMID.2026.v16.i1.1 AMA (American Medical Association) Style Alghazeer R, Elghmasi S, Varbanov M, Philippot S, Hawisa S, Sidati M, Lwaleed B, Azwai S, Eldaghayes I, Gammoudi F, Garbaj A, Alkasak M, Althaluti E. Cytotoxic, anti-coronavirus, and antioxidant activity of Hypericum triquetrifolium: Potential therapeutic applications. J Microbiol Infect Dis. 2026; 16(1): 1-11. doi:10.5455/JMID.2026.v16.i1.1 Vancouver/ICMJE Style Alghazeer R, Elghmasi S, Varbanov M, Philippot S, Hawisa S, Sidati M, Lwaleed B, Azwai S, Eldaghayes I, Gammoudi F, Garbaj A, Alkasak M, Althaluti E. Cytotoxic, anti-coronavirus, and antioxidant activity of Hypericum triquetrifolium: Potential therapeutic applications. J Microbiol Infect Dis. (2026), [cited January 25, 2026]; 16(1): 1-11. doi:10.5455/JMID.2026.v16.i1.1 Harvard Style Alghazeer, R., Elghmasi, . S., Varbanov, . M., Philippot, . S., Hawisa, . S., Sidati, . M., Lwaleed, . B., Azwai, . S., Eldaghayes, . I., Gammoudi, . F., Garbaj, . A., Alkasak, . M. & Althaluti, . E. (2026) Cytotoxic, anti-coronavirus, and antioxidant activity of Hypericum triquetrifolium: Potential therapeutic applications. J Microbiol Infect Dis, 16 (1), 1-11. doi:10.5455/JMID.2026.v16.i1.1 Turabian Style Alghazeer, Rabia, Sana Elghmasi, Mihayl Varbanov, Stéphanie Philippot, Samia Hawisa, Maamar Sidati, Bashir Lwaleed, Salah Azwai, Ibrahim Eldaghayes, Fatim Gammoudi, Aboubaker Garbaj, Musa Alkasak, and Ezdehar Althaluti. 2026. Cytotoxic, anti-coronavirus, and antioxidant activity of Hypericum triquetrifolium: Potential therapeutic applications. Journal of Microbiology and Infectious Diseases, 16 (1), 1-11. doi:10.5455/JMID.2026.v16.i1.1 Chicago Style Alghazeer, Rabia, Sana Elghmasi, Mihayl Varbanov, Stéphanie Philippot, Samia Hawisa, Maamar Sidati, Bashir Lwaleed, Salah Azwai, Ibrahim Eldaghayes, Fatim Gammoudi, Aboubaker Garbaj, Musa Alkasak, and Ezdehar Althaluti. "Cytotoxic, anti-coronavirus, and antioxidant activity of Hypericum triquetrifolium: Potential therapeutic applications." Journal of Microbiology and Infectious Diseases 16 (2026), 1-11. doi:10.5455/JMID.2026.v16.i1.1 MLA (The Modern Language Association) Style Alghazeer, Rabia, Sana Elghmasi, Mihayl Varbanov, Stéphanie Philippot, Samia Hawisa, Maamar Sidati, Bashir Lwaleed, Salah Azwai, Ibrahim Eldaghayes, Fatim Gammoudi, Aboubaker Garbaj, Musa Alkasak, and Ezdehar Althaluti. "Cytotoxic, anti-coronavirus, and antioxidant activity of Hypericum triquetrifolium: Potential therapeutic applications." Journal of Microbiology and Infectious Diseases 16.1 (2026), 1-11. Print. doi:10.5455/JMID.2026.v16.i1.1 APA (American Psychological Association) Style Alghazeer, R., Elghmasi, . S., Varbanov, . M., Philippot, . S., Hawisa, . S., Sidati, . M., Lwaleed, . B., Azwai, . S., Eldaghayes, . I., Gammoudi, . F., Garbaj, . A., Alkasak, . M. & Althaluti, . E. (2026) Cytotoxic, anti-coronavirus, and antioxidant activity of Hypericum triquetrifolium: Potential therapeutic applications. Journal of Microbiology and Infectious Diseases, 16 (1), 1-11. doi:10.5455/JMID.2026.v16.i1.1 |