| Research Article | ||
J. Microbiol. Infect. Dis., (2026), Vol. 16(1): 12–17 Research Article Time to toxin coverage in necrotizing skin infections and associated outcomesTaylor Raiger1, Alison Orvin1,2 and Christian Caveness1*1Department of Pharmacy, WakeMed Health & Hospitals, Raleigh, NC, USA 2UNC Eshelman School of Pharmacy, Chapel Hill, NC, USA *Corresponding Author: Christian Caveness. Department of Pharmacy, WakeMed Health & Hospitals, Raleigh, NC, USA. Email: ccaveness [at] wakemed.org Submitted: 02/09/2025 Revised: 22/11/2025 Accepted: 02/12/2025 Published: 01/01/2026 © 2026 Open Veterinary Journal
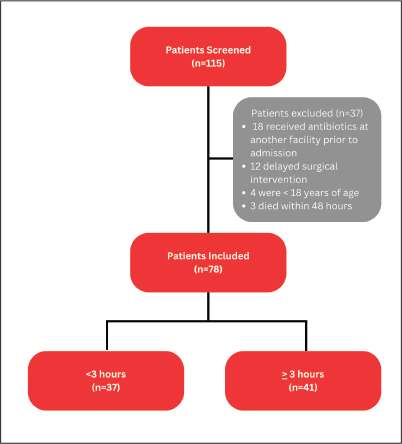
ABSTRACTBackground: Necrotizing soft tissue infections (NSTIs) and the associated exotoxins are associated with a high risk of mortality. Toxin coverage has been shown to decrease mortality, but the appropriate time for toxin coverage is unknown. Aim: The aim of this study is to evaluate whether starting early (<3 hours) toxin coverage improves outcomes compared to late (>3 hours) toxin coverage in relation to the initiation of broad-spectrum antibiotics. Methods: This retrospective, single-center, cohort study compared 30-day all-cause mortality in patients treated with early versus late toxin coverage for necrotizing skin infections. Adult patients admitted between January 2015 and December 2024 who had a diagnosis code of necrotizing fasciitis and were administered IV or PO clindamycin or linezolid within 72 hours of broad-spectrum antibiotics were included. The primary outcome was all-cause mortality at 30 days. Secondary outcomes included intensive care unit (ICU) length of stay (LOS), ICU-free days at 30 days, hospital LOS, acute kidney injury (AKI), Clostridioides difficile infection (CDI), serotonin syndrome, and thrombocytopenia. Results: Baseline characteristics were similar between groups. Both groups had a similar number of ICU admissions (78.4% vs. 75.6%), Charlson Comorbidity Index median of 3 interquartile range (IQR 0-6 vs. 0-7), and Quick Sequential Organ Failure Assessment median of 0 (IQR 0-2 vs. 0-2). Lower extremity was the most common location of infection (29.7% vs. 36.6%), and polymicrobial infections were the most common. Group A Streptococcus accounted for three infections in each group. There was no difference in all-cause mortality at 30 days (5.4% vs. 14.6%, p=0.27). No differences were found in the secondary outcomes of ICU LOS, ICU-free days at 30 days, hospital LOS, rate of AKI, and thrombocytopenia. There was no incidence of serotonin syndrome and a single incidence of CDI in the early toxin coverage group. Conclusion: In this retrospective study, there was no statistically significant difference between 30-day all-cause mortality in patients who received toxin coverage in <3 hours compared to >3 hours in relation to broad-spectrum antibiotics for the treatment of NSTIs. Further studies with a larger study population are needed to determine if the time to toxin coverage impacts mortality. Keywords: Antibiotics, Mortality, NSTI. IntroductionNecrotizing soft-tissue infections (NSTIs) are characterized by a fulminant clinical course with rapidly spreading infection and extensive necrosis of the skin and associated structures (De Waele, 2012). These infections can occur after major or minor traumatic injuries, including both penetrating and nonpenetrating trauma, lacerations, insect bites, obstetric and gynecologic procedures, and post-surgical procedures (Stevens and Bryant, 2017). There are several causes of NSTIs, including polymicrobial and monomicrobial infections, frequently caused by toxin-producing organisms such as Streptococcus pyogenes. Other causative bacteria include methicillin-resistant Staphylococcus aureus (MRSA), Vibrio spp., and Clostridium spp., all of which commonly produce toxins and can precipitate toxic shock (De Waele, 2012; Urbina et al., 2021; Heil et al., 2023). Initial management of NSTIs relies on broad-spectrum antibiotics and rapid surgical debridement. NSTIs and their associated exotoxins significantly increase the risk of mortality, and it has been shown that antibiotics targeting protein synthesis, such as clindamycin or linezolid, can improve mortality in these patients (De Waele, 2012; Urbina et al., 2021; Dorazio et al., 2023). However, the timing of anti-toxin initiation has not been extensively studied. Furthermore, it has been shown that early antimicrobials in sepsis, as well as early surgical debridement in NSTIs improve survival, and we theorize that the same may apply for antibiotics that inhibit toxin production (Urbina et al., 2021; Dorazio et al., 2023). The 3-hour sepsis bundle, which includes obtaining a blood lactate level, blood cultures, administering IVF at 30 ml/kg, and broad-spectrum antibiotics, all within 3 hours of resuscitation, showed a mortality benefit (Evans et al., 2021). Here we examine if receiving toxin coverage either <3 hours or >3 hours from broad-spectrum antibiotics shows comparable results. Therefore, the aim of this study is to evaluate whether starting early (<3 hours) toxin coverage improves outcomes compared to late (>3 hours) toxin coverage in relation to broad-spectrum antibiotic initiation. Materials and MethodsStudy designThis retrospective, single-center, cohort study evaluated the time to toxin coverage with either clindamycin or linezolid for NSTIs at a large community health system in North Carolina. Patients with an inpatient admission from January 2015 to December 2024 were evaluated for inclusion. A list of study patients was obtained using Population Builder™, a SQL database that references data from our electronic medical record, Epic Systems®. Adult patients were included if they were admitted January 2015 to December 2024 with an ICD10 code: M72.6 or ICD 9 code: 728.86, both coding for "Necrotizing fasciitis" and had an administration of IV or PO clindamycin or linezolid within 72 hours of broad-spectrum antibiotics. All patients included were divided into two groups determined by time to toxin coverage as <3 hours or >3 hours from broad-spectrum antibiotics (including vancomycin, cefepime, ceftriaxone, piperacillin-tazobactam, ampicillin-sulbactam, and/or meropenem for empiric coverage). Patients were excluded if they received an antibiotic at another facility prior to admission, had delayed surgical intervention (defined as residing in the emergency department for more than 24 hours or the patient refusing surgical intervention), and death within 48 hours of admission. OutcomesThe primary outcome was all-cause mortality at 30 days. Secondary outcomes included intensive care unit (ICU) length of stay (LOS), ICU-free days at 30 days, hospital LOS, rates of acute kidney injury (AKI), Clostridium difficile infection (CDI), thrombocytopenia, and serotonin syndrome. Data collectionData were obtained from the electronic medical record and included demographic data, culture results, timing of surgical intervention, and administration of antibiotics. The Charlson Comorbidity Index (CCI), quick sequential organ failure assessment (qSOFA), and systemic inflammatory response syndrome criteria (SIRS) were calculated based on physician assessment and laboratory values upon admission. Empiric antibiotic regimens were recorded based on the time the first antibiotic was administered. At least one administration of clindamycin or linezolid was confirmed by the medication administration record (MAR) and timing of administration was recorded. Information collected in Microsoft Forms® was exported into a secured Excel® spreadsheet. Adverse drug events that were monitored included thrombocytopenia defined as platelets less than 150 k/ul, serotonin syndrome (determined by Hunter Criteria), CDI infection (determined by positive antigen-toxin test), and AKI (defined by KDIGO guidelines). For patients on vasopressors, the number, duration, and agent(s) were recorded based on MAR administration records. Corticosteroid and intravenous immunoglobulin (IVIG) use was also recorded using MAR administrations. The number of patients who had an infectious diseases (IDs) consult was also collected. Statistical analysisContinuous variables were evaluated using the Wilcoxon rank-sum test and described using median and interquartile range (IQR). Categorical data was assessed using chi square test and the Fisher exact test. These variables were described using frequencies (%). A p-value of <0.05 was used to determine statistical significance. Finally, a Log-Rank test was performed to create a Kaplan Meier curve to illustrate portion of patients surviving. All statistical analyses were performed using SAS JMP® software, version 9 (SAS Institute, Cary, NC). Ethical approvalThis study received exempt status from our institutional review board due to its retrospective study design. ResultsOne hundred and fifteen patients were identified using the diagnosis codes for "Necrotizing fasciitis" and had an administration of IV or PO clindamycin or linezolid within 72 hours of broad-spectrum antibiotics during their encounter. Seventy-eight met the inclusion criteria, with 37 patients (47%) receiving toxin coverage in <3 hours and 41 patients (53%) receiving toxin coverage in >3 hours of broad-spectrum antibiotics. The most common reason for exclusion was receipt of antibiotics at another facility prior to admission (Fig. 1).
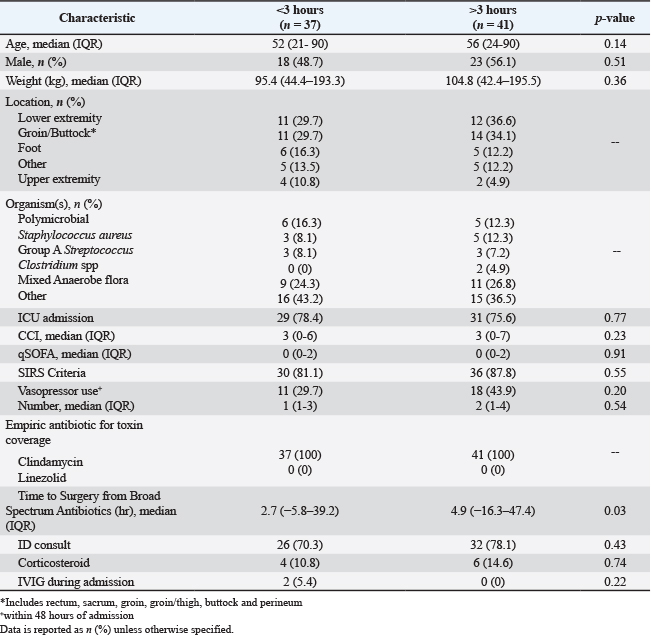
Figure 1. Study population. Baseline characteristics were similar between groups (Table 1). Both groups had a similar number of ICU admissions (78.4% vs. 75.6%), CCI median of 3 (IQR 0-6 vs. 0-7), and qSOFA median of 0 (IQR 0-2 vs. 0-2). Lower extremity was the most common location of infection (29.7% vs. 36.6%), and polymicrobial infections were the most common. Group A Streptococcus accounted for three infections in each group. In the early toxin coverage group, there was a significantly shorter time to surgery from broad-spectrum antibiotics (2.7 vs. 4.9 hours, p=0.03). All patients were empirically started on clindamycin for toxin coverage. Table 1. Baseline characteristics.
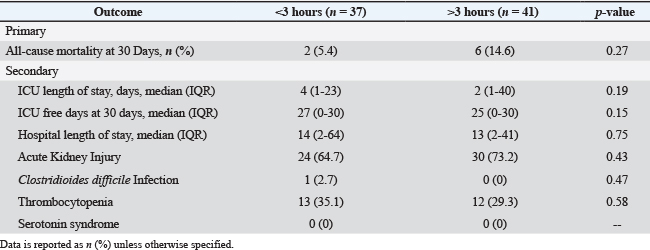
There was no statistically significant difference in all-cause mortality at 30 days (5.4% vs. 14.6%, p=0.27) (Table 2). There were no differences in the secondary outcomes of ICU LOS (4 vs. 2 days, p=0.19), ICU-free days at 30 days (27 days vs. 25 days, p=0.15), hospital LOS (14 vs. 13 days, p=0.75), and rate of AKI (24 vs. 30 patients, p=0.43). The secondary safety outcome of thrombocytopenia occurred similarly between groups (35.1% vs. 29.3%, p=0.58). There was no incidence of serotonin syndrome and a single incidence of CDI in the early toxin coverage group. Table 2. Outcomes.
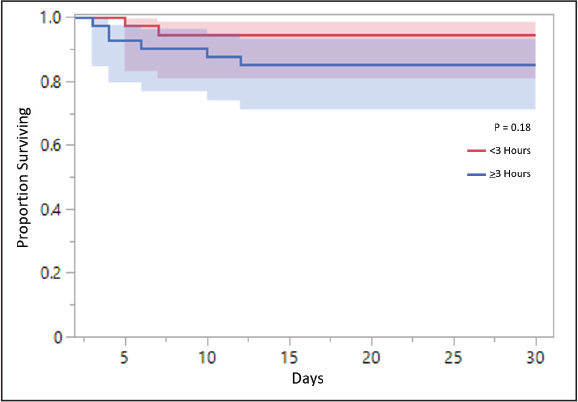
A survivorship curve was generated to better visualize overall survival within the 30-day interval between the early and late toxin coverage groups, which correlated to a non-statistically significant difference (p=0.18) (Fig. 2).
Figure 2. Survivorship between early versus late toxin coverage. DiscussionIn this retrospective, single-center, cohort study examining the impact of early (<3 hours) versus late (>3 hours) time to toxin coverage from broad spectrum antibiotics in patients with a NSTI at a large community hospital, the primary outcome of all-cause mortality at 30 days was not statistically significant. NSTIs, specifically those caused by toxin-producing organisms such as S. pyogenes, MRSA, Vibrio spp., and Clostridium spp. are associated with high mortality. As a result, studies have established that early surgical debridement improves mortality with the surviving sepsis guidelines also emphasizing the importance of the 3-hour sepsis bundle to improve mortality (Gelbard et al., 2018; Evans et al., 2021). Babiker et al. (2021)evaluated beta-lactam plus clindamycin initiation within 72 hours of positive cultures growing beta-hemolytic Streptococcus compared to beta-lactam alone and found a significant reduction in in-hospital mortality (6.5% vs. 11%, p=0.042). We found similar rates of mortality in our two groups (5.4% vs. 14.6%, p=0.27). However, they only found a statistically significant reduction in mortality in patients with invasive group A Streptococcus (GAS) infections, whereas we only had three patients in each of our groups with a documented GAS infection. They did not report time to surgery, which could have confounded their outcomes. There have been only small retrospective studies looking at the early addition of adjunct clindamycin. Hamada et al. (2022) investigated patients with a diagnosis of “sepsis due to group A Streptococcus” and “necrotizing fasciitis” who received clindamycin on the day of admission compared to those that did not receive clindamycin. They found that clindamycin was not associated with a reduction in hospital mortality (19.2% vs. 17.5%, p=0.74) (Hamada et al., 2022). While we found lower rates of mortality and no statistically significant difference, all our patients received clindamycin whether early or late in comparison to broad-spectrum antibiotics and we only had a small number of patients in each group with a GAS infection. This study addresses the gap of if early anti-toxin initiation is advantageous as previous studies explored the addition of toxin covering agents within 24–72 hours of admission or of culture collection. The strengths of this study include well-balanced groups, wide variety of infection sites and almost all patients receiving appropriate toxin coverage within 24 hours. Limitations of this study include a small sample size, thereby, not allowing power to be met, and the potential for a possible difference that could not be detected. The retrospective nature, low rate of Group A Streptococcus, and inability to account for differences in surgical management are further limitations. Finally, due to the lack of empiric use of linezolid at our institution, the impact of early linezolid on mortality could not be ascertained. Potential confounders within the study include time to surgery from hospital presentation, as well as recognition of NSTI, which can vary between patients. Furthermore, the time to surgery from broad-spectrum antibiotics was different between groups and statistically significant. Given that the early group had a shorter time to surgery, this may have decreased their mortality. ConclusionIn this small, retrospective cohort, single center study, we did not find a statistically significant difference between all-cause mortality at 30 days in patients who received toxin coverage in <3 hours compared to >3 hours in relation to broad-spectrum antibiotics for the treatment of NSTIs. There was no difference in ICU LOS, ICU-free days at 30 days, hospital LOS, AKI, CDI, thrombocytopenia, or serotonin syndrome rates between groups. Further studies with a larger study population, with a larger proportion of toxin-producing organisms, are needed to determine if the time to toxin coverage impacts mortality. AcknowledgmentsThe authors have no acknowledgments. Conflict of interestThe authors declare that there is no conflict of interest. FundingThis research received no specific grant. Authors’ contributionsInvestigations: Taylor Raiger, Christian Caveness. Data analysis: Taylor Raiger, Alison Orvin. Draft writing: Taylor Raiger and Edits: Christian Caveness, Alison Orvin.All authors approved the final version. Data availabilityAll data are provided in the manuscript. ReferencesBabiker, A., Li, X., Lai, Y.L., Strich, J.R., Warner, S., Sarzynski, S., Dekker, J.P., Danner, R.L. and Kadri, S.S. 2021. Effectiveness of adjunctive clindamycin in β-lactam antibiotic-treated patients with invasive β-haemolytic streptococcal infections in US hospitals: a retrospective multicentre cohort study. Lancet. Infect. Dis. 21, 697–710. De Waele, J.J. 2012. Management of necrotizing skin and soft tissue infections. Expert Rev. Anti. Infect. Ther. 10, 805–814. Dorazio, J., Chiappelli, A.L., Shields, R.K., Tsai, Y.V., Skinker, P., Nabozny, M.J., Bauza, G., Forsythe, R., Rosengart, M.R., Gunn, S.R., Marini, R., Clarke, L., Falcione, B., Ludwig, J. and Mccreary, E.K. 2023. Clindamycin Plus Vancomycin Versus Linezolid for Treatment of Necrotizing Soft Tissue Infection. Open. Forum. Infect. Dis. 10(10), 258. Evans, L., Rhodes, A., Alhazzani, W., Antonelli, M., Coopersmith, C.M., French, C., Machado, F.R., Mcintyre, L., Ostermann, M., Prescott, H.C., Schorr, C., Simpson, S., Wiersinga, W.J., Alshamsi, F., Angus, D.C., Arabi, Y., Azevedo, L., Beale, R., Beilman, G., Belley-Cote, E., Burry, L., Cecconi, M., Centofanti, J., Coz Yataco, A., De Waele, J., Dellinger, R.P., Doi, K., Du, B., Estenssoro, E., Ferrer, R., Gomersall, C., Hodgson, C., Møller, M.H., Iwashyna, T., Jacob, S., Kleinpell, R., Klompas, M., Koh, Y., Kumar, A., Kwizera, A., Lobo, S., Masur, H., Mcgloughlin, S., Mehta, S., Mehta, Y., Mer, M., Nunnally, M., Oczkowski, S., Osborn, T., Papathanassoglou, E., Perner, A., Puskarich, M., Roberts, J., Schweickert, W., Seckel, M., Sevransky, J., Sprung, C.L., Welte, T., Zimmerman, J. and Levy, M. 2021. Surviving sepsis campaign: international guidelines for management of sepsis and septic shock 2021. Intensive. Care. Med. 47, 1181–1247. Gelbard, R.B., Ferrada, P., Yeh, D.D., Williams, B.H., Loor, M., Yon, J., Mentzer, C., Khwaja, K., Khan, M.A., Kohli, A., Bulger, E.M. and Robinson, B.R.H. 2018. Optimal timing of initial debridement for necrotizing soft tissue infection: a Practice Management Guideline from the Eastern Association for the Surgery of Trauma. J. Trauma Acute Care Surg. 85, 208–214. Hamada, S., Nakajima, M., Kaszynski, R.H., Otaka, S., Goto, H., Matsui, H., Fushimi, K., Yamaguchi, Y. and Yasunaga, H. 2022. Association between adjunct clindamycin and in-hospital mortality in patients with necrotizing soft tissue infection due to group A Streptococcus: a nationwide cohort study. Eur. J. Clin. Microbiol. Infect. Dis. 41, 263–270. Heil, E.L., Kaur, H., Atalla, A., Basappa, S., Mathew, M., Seung, H., Johnson, J.K. and Schrank, G.M. 2023. Comparison of Adjuvant Clindamycin vs Linezolid for Severe Invasive Group A Streptococcus Skin and Soft Tissue Infections. Open Forum Infect. Dis. 10, 588. Stevens, D.L. and Bryant, A.E. 2017. Necrotizing Soft-Tissue Infections. N. Engl. J. Med. 377, 2253–2265. Urbina, T., Razazi, K., Ourghanlian, C., Woerther, P.L., Chosidow, O., Lepeule, R. and De Prost, N. 2021. Antibiotics in Necrotizing Soft Tissue Infections. Antibiotics (Basel). 10, 1104. | ||
| How to Cite this Article |
| Pubmed Style Raiger T, Orvin A, Caveness C. Time to toxin coverage in necrotizing skin infections and associated outcomes. J Microbiol Infect Dis. 2026; 16(1): 12-17. doi:10.5455/JMID.2026.v16.i1.2 Web Style Raiger T, Orvin A, Caveness C. Time to toxin coverage in necrotizing skin infections and associated outcomes. https://www.jmidonline.org/?mno=281536 [Access: January 25, 2026]. doi:10.5455/JMID.2026.v16.i1.2 AMA (American Medical Association) Style Raiger T, Orvin A, Caveness C. Time to toxin coverage in necrotizing skin infections and associated outcomes. J Microbiol Infect Dis. 2026; 16(1): 12-17. doi:10.5455/JMID.2026.v16.i1.2 Vancouver/ICMJE Style Raiger T, Orvin A, Caveness C. Time to toxin coverage in necrotizing skin infections and associated outcomes. J Microbiol Infect Dis. (2026), [cited January 25, 2026]; 16(1): 12-17. doi:10.5455/JMID.2026.v16.i1.2 Harvard Style Raiger, T., Orvin, . A. & Caveness, . C. (2026) Time to toxin coverage in necrotizing skin infections and associated outcomes. J Microbiol Infect Dis, 16 (1), 12-17. doi:10.5455/JMID.2026.v16.i1.2 Turabian Style Raiger, Taylor, Alison Orvin, and Christian Caveness. 2026. Time to toxin coverage in necrotizing skin infections and associated outcomes. Journal of Microbiology and Infectious Diseases, 16 (1), 12-17. doi:10.5455/JMID.2026.v16.i1.2 Chicago Style Raiger, Taylor, Alison Orvin, and Christian Caveness. "Time to toxin coverage in necrotizing skin infections and associated outcomes." Journal of Microbiology and Infectious Diseases 16 (2026), 12-17. doi:10.5455/JMID.2026.v16.i1.2 MLA (The Modern Language Association) Style Raiger, Taylor, Alison Orvin, and Christian Caveness. "Time to toxin coverage in necrotizing skin infections and associated outcomes." Journal of Microbiology and Infectious Diseases 16.1 (2026), 12-17. Print. doi:10.5455/JMID.2026.v16.i1.2 APA (American Psychological Association) Style Raiger, T., Orvin, . A. & Caveness, . C. (2026) Time to toxin coverage in necrotizing skin infections and associated outcomes. Journal of Microbiology and Infectious Diseases, 16 (1), 12-17. doi:10.5455/JMID.2026.v16.i1.2 |